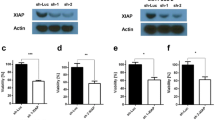
Abstract
Imexon is an aziridine-containing small molecule currently in Phase I clinical trials. This agent has been shown to bind to thiols and increase intracellular oxidants, inducing apoptosis in hematologic cancer cells. Pancreatic cancers are known to be sensitive to oxidation, suggesting this disease may be an appropriate target for this agent. The current report examines the activity of imexon in pancreatic cells. Imexon induced concentration-dependent and time-dependent apoptosis in a panel of six human pancreatic carcinoma cell (PCC) lines. The mean IC50 (SD) for growth inhibition by the SRB assay was 200 (101) µM for a 48 h exposure with a range of 64–358 µM. Cell killing was schedule-dependent, favoring exposure times ≥48 h. Imexon-treated MiaPaCa-2 cells underwent non-lethal growth arrest following exposure to concentrations ≤200 µM for 48 h. When concentrations were increased to 300 µM for ≥48 h, the MiaPaCa-2 cells arrested in G2 phase and activated caspases 3, 8, and 9 were detected. After a 72 h exposure to the IC80 concentration of imexon, cells exhibited a loss of mitochondrial membrane potential detected by CMXRos staining. However, there was no loss of reduced cellular thiols unless very high concentrations of ≥400 µM were used. In contrast, reactive oxygen species (ROS) were elevated in a dose-dependent fashion, starting at very low imexon concentrations. Imexon also significantly inhibited MiaPaCa-2 tumor growth in SCID mice at 100 mg/kg/d for 9 d. The tumor growth inhibition (% T/C) was 27% of control, and the tumor growth delay was 21 d, indicating an active agent by NCI standards.
The levels of imexon that are cytotoxic in human PCC’s are achievable based on the preliminary results of the ongoing Phase I trial. Imexon appears to be active against PCCs in vitro and has an entirely novel mechanism of action involving G2 arrest, accumulation of ROS, and the induction of apoptosis.
Similar content being viewed by others
References
Evans DB, Abbruzzese JL, Rich TA. Cancer of the pancreas, in Cancer: Principles and Practice of Oncology, DeVita VT Jr, Hellman S, Rosenberg SA, eds. Lippincott-Raven, Philadelphia, PA, 1997; pp. 1054–1087.
Jemal A, Murray T, Samuels A, Ghafoor A, Ward E, Thun MJ. CA Cancer J Clin 2003;53:5–26.
Burris III HA, Moore MJ, Andersen J, et al. Improvements in survival and clinical benefit with gemcitabine as first-line therapy for patients with advanced pancreas cancer: a randomized trial. J Clin Oncol 1997;15:2403–2413.
Conroy T, Paillot B, Francois E, et al. Final results of folfirinox: a triple combination of 5-FU/leucovorin, irinotecan, and oxaliplatin as first line chemotherapy in advanced pancreatic adenocarcinoma [Abstr]. Proc Am Soc Clin Oncol 2003;22:261.
Philip PA, Zalupski MM, Vaitkevicius VK, et al. Phase II study of gemcitabine and cisplatin in the treatment of patients with advanced pancreatic carcinoma. Cancer 2001;92:569–577.
El-Rayes BF, Zalupski MM, Shields AF, et al. Phase II study of gemcitabine, cisplatin, and infusional fluorouracil in advanced pancreatic cancer. J Clin Oncol 2003;21:2920–2925.
Reni M, Passoni P, Panucci MG, et al. Definitive results of a phase II trial of cisplatin, epirubicin, continuous-infusion fluorouracil, and gemcitabine in stage IV pancreatic adenocarcinoma. J Clin Oncol 2001;19:2679–2686.
Fine RL, Fogelman DR, Sherman W, et al. The GTX regimen: a biochemically synergistic combination for advanced pancreatic cancer [Abstr]. Proc Am Soc Clin Oncol 2003;22:281.
Xiong HQ, Rosenberg A, LoBuglio A, et al. Cetuximab, a monoclonal antibody targeting the epidermal growth factor receptor, in combination with gemcitabine for advanced pancreatic cancer: a multicenter phase II trial. J Clin Oncol 2004;22:2610–2616.
Cohen SL, Ho L, Ranganathan S, et al. Phase II and pharmacodynamic study of the farnesyltransferase inhibitor R115777 as initial therapy in patients with metastatic pancreatic adenocarcinoma. J Clin Oncol 2003;21:1301–1306.
Lersch C, Van Cutsem J, Armado R, et al. Randomized Phase II study of SCH 66336 and gemcitabine in the treatment of metastatic adenocarcinoma of the pancreas [Abstr]. Proc Am Soc Clin Oncol 2001;20:153a.
Moore MJ, Hamm J, Dancey J, et al., and the National Cancer Institute of Canada Clinical Trials Group. Comparison of gemcitabine versus the matrix metalloproteinase inhibitor Bay 12-9566 in patients with advanced or metastatic adenocarcinoma of the pancreas: a Phase III trial of the National Cancer Institute of Canada Clinical Trials Group. J Clin Oncol 2003;21:3296–3302.
Bramhall SR, Schulz J, Nemunaitis J, Brown PD, Baillet M, Buckels JA. A double-blind placebo-controlled, randomized study comparing gemcitabine and marimastat with gemcitabine and placebo as first line therapy in patients with advanced pancreatic cancer. Br J Cancer 2002;87:161–167.
Appleman LJ, Ryan DP, Clark JW, et al. Phase I dose escalation study of bortezomib and gemcitabine safety and tolerability in patients with advanced solid tumors [Abstr]. Proc Am Soc Clin Oncol 2003;22:209.
Kemp KR, Liehr JG, Giovanella B. Combined radiation and 9-nitrocamptothecin (Rubitecan) in the treatment of locally advanced pancreatic cancer. Ann NY Acad Sci 2000;922:320–323.
Dvorakova K, Payne CM, Tome M, Briehl M, McClure T, Dorr RT. Induction of oxidative stress and apoptosis in meyloma cells by imexon. Biochem Pharmacol 2000;60:749–758.
Dvorakova K, Waltmire CN, Payne CM, Tome ME, Briehl MM, Dorr RT. Induction of mitochondrial changes in myeloma cells by imexon. Blood 2000;97:3544–3550.
Dvorakova K, Payne CM, Landowski TH, Tome ME, Halperin DS, Dorr RT. Imexon activates an Intrinsic apoptosis pathway in RPM18226 myeloma cells. Anticancer Drugs 2002;13:1031–1042.
Evens AM, Prachand S, Shi B, Paniaqua M, Gordon LI, Gartenhaus RB. Imexon-induced apoptosis in multiple myeloma tumor cells in caspase-8 dependent. Clin Cancer Res 2004;10:1481–1491.
Salmon SE, Hersh EM. Sensitivity of multiple myeloma to imexon in the human tumor cloning assay. J Natl Cancer Inst 1994;86:228–230.
Cullen JJ, Mitros FA, Oberley LW. Expression of antioxidant enzymes in diseases of the human pancreas: another link between chronic pancreatitis and pancreatic cancer. Pancreas 2003;26:23–27.
Liu J, Hinkhouse MM, Sun W, et al. Redox regulation of pancreatic cancer cell growth: role of glutathione peroxidase in the suppression of the malignant phenotype. Hum Gene Ther 2004;15:239–250.
Kadlubar FF, Anderson KE, Haussermann S, et al. Comparison of DNA adduct levels associated with oxidative stress in human pancreas. Mutat Res 1998;405:125–133.
Wang M, Abbruzzese JL, Friess H, et al. DNA adducts in human pancreatic tissues and their potential role in carcinogenesis. Cancer Res 1998;58:38–41.
Schnelldorfer T, Gansauge S, Gansauge F, Schlosser S, Beger HG, Nussler AK. Glutathione depletion causes cell growth inhibition and enhanced apoptosis in pancreatic cancer cells. Cancer 2000;89:1440–1447.
Loor R, Nowa, NJ, Manzo ML, Douglass HO, Chu TM. Use of pancreas-specific antigen in immunodiagnosis of pancreatic cancer. Clin Lab Med 1982;2:567–578.
Schoumacher RA, Ram J, Iannuzzi MC, et al. Acystic fibrosis pancreatic adenocarcinoma cell line. Proc Natl Acad Sci USA 1990;87:4012–4016.
Yunis AA, Arimura GK, Russin DJ. Human pancreatic cancinoma (MIA PaCa-2) in continuous culture. Sensitivity to asparaginase. Int J Cancer 1977;19:128–135.
Lieber M, Mazzetta J, Nelson-Rees W, Kaplan M, Todaro G. Establishment of a continuous tumor-cell line (panc-1) from a human carcinoma of the exocrine pancreas. Int J Cancer 1975;15:741–747.
Drucker BJ, Marincola FM, Siao DY, Donlon TA, Bangs CD, Holder WD Jr. A new human pancreatic carcinoma cell line developed for adoptive immunotherapy studies with lymphokine-activated killer cells in nude mice. In Vitro Cell Dev Biol 1988;24:1179–1187.
Korc M, Meltzer P, Trent J. Enhanced expression of epidermal growth factor receptor correlates with alterations of chromosome 7 in human pancreatic cancer. Proc Nat Acad Sci USA 1986;83:5141–5144.
Skehan P, Storeng R, Scudiero D, et al. New colorimetric cytotoxicity assay for anticancer-drug screening. J Natl Can Inst 1990;82:1107–1112.
Gavrieli Y, Sherman Y, Ben-Sasson SA. Identification of programmed cell death in situ via specific labeling of nuclear DNA fragmentation. J Cell Biol 1992;119:493–501.
Poot M, Kavanagh TJ, Kang HC, Haugland RP, Rabinovitch PS. Flow cytometric analysis of cell cycle-dependent changes in cell thiol level by combining a new laser dye with Hoechst 33342. Cytometry 1991;12:184–187.
Anderson ME. Enzymatic and chemical methods for the determination of Glutathione, in Glutathione: Chemical, Biochemical and Medical Aspects. Vol. A., Dolphin D, Poulson R, Avramovic O, eds. John Wiley and Sons, New York, NY, 1989; pp. 339–365
Macho A, Decaudin D, Castedo M, et al. Chloromethyl-X-Rosamine is an aldehyde-fixable potential-sensitive fluorochrome for the detection of early apoptosis. Cytometry 1996;25:333–340.
Mancini M, Anderson BO, Caldwell E, Sedghinasab M, Paty PB, Hockenbery DM. Mitochondrial proliferation and paradoxical membrane depolarization during terminal differentiation and apoptosis in a human colon carcinoma cell line. J Cell Biol 1997;138:449–469
Carter WO, Narayanan PK, Robinson JP. Intracellular hydrogen peroxide and superoxide anion detection in endothelial cells. J Leukoc Biol 1994;55:253–258.
Krishan A. Rapid cytoflurorometric analysis of mammalian cell cycle by propidium iodide staining. J Cell Biol 1975;66:188–193
Amstad PA, Yu G, Johnson GL, Lee BW, Dhawan S, Phelps DJ. Detection of caspase activation in situ by fluorochromelabeled caspase inhibitors. Biotechniques 2001;31:608–610.
Bissery MC, Guenard D, Gueritte-Voegelein F, Lavelle F. Experimental antitumor activity of taxotere (RP 56976, NSC 628503), a taxol analogue. Cancer Res 1991;51:4845–4852.
Iyengar BS, Dorr RT, Remers WA. Chemical basis for the biological activity of imexon and related cyanoaziridines. J Med Chem 2004;47:218–223.
Dragovich T, Mendelsohn D, Gordon M, et al. Phase I trial of imexon: (IMX) in patients (pts) with advanced solid tumors [Abstr 3153]. Proc Amer Soc Clin Oncol Mtg 2005;23:229.
Nakamura H, Bai J, Nishinaka Y, et al. Expression of thioredoxin and glutaredoxin, redox-regulating proteins, in pancreatic cancer. Can Detect Prev 2000;24:53–60.
Cullen JJ, Hinkhouse MM, Grady M, et al. Dicumarol inhibition of NADPH: Quinone oxidoreductase induces growth inhibition of pancreatic cancer via a superoxide-mediated mechanism. Cancer Res 2003;63:5513–5520.
Cullen JJ, Weydert C, Hinkhouse MM, et al. The role of manganese superoxide dismutase in the growth of pancreatic adenocarcinoma. Cancer Res 2003;63:1297–1303.
Irani K, Xia Y, Zweier JL, et al. Mitogenic signaling mediated by oxidants in Ras-transformed fibroblasts. Science 1997;275:1649–1652.
Chandel NS, McClintock DS, Feliciano CE, et al. Reactive oxygen species generated at mitochondrial complex III stabilize hypoxia-inducible factor-1 alpha during hypoxia. J Biol Chem 2000;275:25,130–25,138.
Yang J-Q, Li S, Domann FF, Buettner GR, et al. Superoxide generation in v-Ha-ras-transduced human keratinocyte HaCaT cells. Mol Carcinog 1999;26:180–188.
Motojima K, Urano T, Nagata Y, et al. Detection of point mutations in the Kirsten-ras oncogene provides evidence for the multicentricity of pancreatic carcinoma. Ann Surg 1993;217:138–143.
Motojima K, Tsunoda T, Kanematsu T, et al. Distinguishing pancreastic carcinoma from other periampullary carcinomas by analysis of mutations in the Kirsten-ras oncogene. Ann Surg 1991;214:657–662.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Dorr, R.T., Raymond, M.A., Landowski, T.H. et al. Induction of apoptosis and cell cycle arrest by imexon in human pancreatic cancer cell lines. Int J Gastrointest Canc 36, 15–28 (2005). https://doi.org/10.1385/IJGC:36:1:015
Issue Date:
DOI: https://doi.org/10.1385/IJGC:36:1:015