Abstract
Background
Hypoxia inducible factor-1α (HIF-1α) is a major regulator of tumorigenesis in hypoxic conditions and therefore represents a potential therapeutic target in colorectal cancer (CRC). Clinical significance of HIF-1α expression in liver metastases has not been elucidated. Therefore, this study aimed to clarify the clinical significance of HIF-1α expression in colorectal liver metastasis (CRLM).
Methods
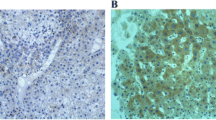
We retrospectively analyzed 64 patients who underwent curative resection of CRLM from 2000 to 2008. We evaluated HIF-1α expression by immunohistochemical staining and analyzed its association with several clinicopathological characteristics, including vascular endothelial growth factor (VEGF) expression. We analyzed the mutation status of genes involved in CRC (p53, KRAS, BRAF, and PIK3CA). Finally, we compared HIF-1α expression between the primary tumor and the corresponding liver metastases.
Results
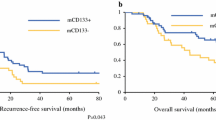
We found a significant positive correlation between HIF-1α expression in liver metastases and PIK3CA mutation status (p = 0.019). A significant correlation was also observed between the expressions of HIF-1α and VEGF in liver metastases and primary tumors (p = 0.015, 0.024, respectively). High HIF-1α expression in liver metastases was an independent risk factor for recurrence (p = 0.031).
Conclusions
Our results suggest a possible induction of HIF-1α expression by mutant PIK3CA. The expressions of HIF-1α and VEGF in liver metastases significantly correlated with those in the corresponding primary tumor. Overexpression of HIF-1α was an independent risk factor for recurrence after curative resection of CRLM, suggesting that HIF-1α represents an important candidate for the treatment of CRLM in a subset of patients with high HIF-1α expression.


Similar content being viewed by others
References
Simmonds PC, Primrose JN, Colquitt JL, Garden OJ, Poston GJ, Rees M. Surgical resection of hepatic metastases from colorectal cancer: a systematic review of published studies. Br J Cancer. 2006;94:982–999.
Choti MA, Sitzmann JV, Tiburi MF, et al. Trends in long-term survival following liver resection for hepatic colorectal metastases. Ann Surg. 2002;235:759–766.
Assumpcao L, Choti MA, Gleisner AL, et al. Patterns of recurrence following liver resection for colorectal metastases: effect of primary rectal tumor site. Arch Surg. 2008;143:743–749.
Semenza GL. Targeting HIF-1 for cancer therapy. Nat Rev Cancer. 2003;3:721–732.
Semenza GL. Defining the role of hypoxia-inducible factor 1 in cancer biology and therapeutics. Oncogene. 2010;29:625–634.
Rasheed S, Harris AL, Tekkis PP, et al. Hypoxia-inducible factor-1alpha and -2 alpha are expressed in most rectal cancers but only hypoxia-inducible factor-1 alpha is associated with prognosis. Br J Cancer. 2009;100:1666–1673.
Baba Y, Nosho K, Shima K, et al. HIF1A overexpression is associated with poor prognosis in a cohort of 731 colorectal cancers. Am J Pathol. 2010;176:2292–2301.
Kuwai T, Kitadai Y, Tanaka S, et al. Expression of hypoxia-inducible factor-1 alpha is associated with tumor vascularization in human colorectal carcinoma. Int J Cancer. 2003;105:176–181.
Yoshimura H, Dhar DK, Kohno H, et al. Prognostic impact of hypoxia-inducible factors 1alpha and 2alpha in colorectal cancer patients: correlation with tumor angiogenesis and cyclooxygenase-2 expression. Clin Cancer Res. 2004;10:8554–8560.
Cao D, Hou M, Guan YS, et al. Expression of HIF-1alpha and VEGF in colorectal cancer: association with clinical outcomes and prognostic implications. BMC Cancer. 2009;9:432.
Imamura T, Kikuchi H, Herraiz MT, et al. HIF-1alpha and HIF-2alpha have divergent roles in colon cancer. Int J Cancer. 2009;124:763–771.
Mizukami Y, Jo WS, Duerr EM, et al. Induction of interleukin-8 preserves the angiogenic response in HIF-1alpha-deficient colon cancer cells. Nat Med. 2005;11:992–997.
Frede S, Freitag P, Otto T, et al. The proinflammatory cytokine interleukin 1beta and hypoxia cooperatively induce the expression of adrenomedullin in ovarian carcinoma cells through hypoxia inducible factor 1 activation. Cancer Res. 2005;65:4690–4697.
Karhausen J, Haase VH, Colgan SP. Inflammatory hypoxia: role of hypoxia-inducible factor. Cell Cycle. 2005;4:256–258.
van der Bilt JD, Soeters ME, Duyverman AM, et al. Perinecrotic hypoxia contributes to ischemia/reperfusion-accelerated outgrowth of colorectal micrometastases. Am J Pathol. 2007;170:1379–1388.
Moeller BJ, Cao Y, Li CY, et al. Radiation activates HIF-1 to regulate vascular radiosensitivity in tumors: role of reoxygenation, free radicals, and stress granules. Cancer Cell. 2004;5:429–441.
Bardos JI, Ashcroft M. Hypoxia-inducible factor-1 and oncogenic signalling. Bioessays. 2004;26:262–269.
Ravi R, Mookerjee B, Bhujwalla ZM, et al. Regulation of tumor angiogenesis by p53-induced degradation of hypoxia-inducible factor 1alpha. Genes Dev. 2000;14:34–44.
Markowitz SD, Bertagnolli MM. Molecular origins of cancer: molecular basis of colorectal cancer. N Engl J Med. 2009;361:2449–2460.
Dindo D, Demartines N, Clavien PA. Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg. 2004;240:205–213.
Ikeda S, Shimizu Y, Fujimori M, et al. Immunohistochemical and mutational analyses of beta-catenin, Ki-ras, and p53 in two subtypes of colorectal mucinous carcinoma. Clin Cancer Res. 2003;9:5660–5665.
Loupakis F, Pollina L, Stasi I, et al. PTEN expression and KRAS mutations on primary tumors and metastases in the prediction of benefit from cetuximab plus irinotecan for patients with metastatic colorectal cancer. J Clin Oncol. 2009;27:2622–2629.
Zhong H, De Marzo AM, Laughner E, et al. Overexpression of hypoxia-inducible factor 1alpha in common human cancers and their metastases. Cancer Res. 1999;59:5830–5835.
Birner P, Schindl M, Obermair A, et al. Overexpression of hypoxia-inducible factor 1alpha is a marker for an unfavorable prognosis in early-stage invasive cervical cancer. Cancer Res. 2000;60:4693–4696.
Schindl M, Schoppmann SF, Samonigg H, et al. Overexpression of hypoxia-inducible factor 1alpha is associated with an unfavorable prognosis in lymph node–positive breast cancer. Clin Cancer Res. 2002;8:1831–1837.
Sullivan R, Pare GC, Frederiksen LJ, et al. Hypoxia-induced resistance to anticancer drugs is associated with decreased senescence and requires hypoxia-inducible factor-1 activity. Mol Cancer Ther. 2008;7:1961–1973.
Guba M, Seeliger H, Kleespies A, et al. Vascular endothelial growth factor in colorectal cancer. Int J Colorectal Dis. 2004;19:510–517.
Kwon HC, Kim SH, Oh SY, et al. Clinicopathological significance of p53, hypoxia-inducible factor 1alpha, and vascular endothelial growth factor expression in colorectal cancer. Anticancer Res. 2010;30:4163–4168.
Kwon HC, Kim SH, Oh SY, et al. Clinicopathological significance of nuclear factor-kappa B, HIF-1 alpha, and vascular endothelial growth factor expression in stage III colorectal cancer. Cancer Sci. 2010;101:1557–1561.
Kikuchi H, Pino MS, Zeng M, et al. Oncogenic KRAS and BRAF differentially regulate hypoxia-inducible factor-1 alpha and -2 alpha in colon cancer. Cancer Res. 2009;69:8499–8506.
Simiantonaki N, Taxeidis M, Jayasinghe C, et al. Hypoxia-inducible factor 1 alpha expression increases during colorectal carcinogenesis and tumor progression. BMC Cancer. 2008;8:320.
Kuramochi H, Hayashi K, Uchida K, et al. Vascular endothelial growth factor messenger RNA expression level is preserved in liver metastases compared with corresponding primary colorectal cancer. Clin Cancer Res. 2006;12:29–33.
Chen J, Li Q, Wang C, et al. Prognostic significance of c-erbB-2 and vascular endothelial growth factor in colorectal liver metastases. Ann Surg Oncol. 2010;17:1555–1563.
Acknowledgment
We thank Yuko Ishida and Midori Kiyokawa for their expert technical assistance, and the Analysis Center of Life Science, Hiroshima University, for the use of their facilities.
Disclosure
The authors declare no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Shimomura, M., Hinoi, T., Kuroda, S. et al. Overexpression of Hypoxia Inducible Factor-1 Alpha is an Independent Risk Factor for Recurrence After Curative Resection of Colorectal Liver Metastases. Ann Surg Oncol 20 (Suppl 3), 527–536 (2013). https://doi.org/10.1245/s10434-013-2945-2
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-013-2945-2