Abstract
Background: The type IV collagenases/gelatinases matrix metalloproteinase-2 (MMP-2) and -9 (MMP-9) play an important role in cancer invasion and metastasis. In the present study, we measured the expression of mRNAs and enzymatic activities of MMP-9 and -2 in prostate tissues and serum samples from men with or without prostate cancer.
Methods: A total of 44 tissue samples (three from healthy volunteers, 21 from patients with benign prostate hyperplasia, 10 from patients with localized prostate cancer and 10 from patients with metastatic disease) and 71 serum samples were collected (20 from healthy volunteers, 26 from patients with benign prostatic hyperplasia, 10 from patients with localized cancer, 15 from patients with metastatic cancer). The level of mRNA for MMP-2 and -9 was determined by semiquantitative reverse transcriptase-polymerase chain reaction (RT-PCR). The enzymatic activity of MMPs was determined by zymography.
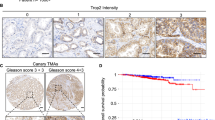
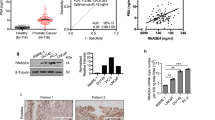
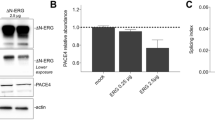
Results: Expression of MMP-9 mRNA was significantly higher in malignant than in nonmalignant prostate tissues (P<0.001), while no significant difference of MMP-2 expression was detected in different prostate tissues. Results of zymography showed that there was significant difference in the enzymatic activity of MMP-9, but not MMP-2, among normal prostate, BPH, localized and metastatic prostate cancer tissues, serum samples (P<0.05). The active form of MMP-2, with a molecular mass of 62 kDa, was detected in normal prostate, BPH and prostate cancer tissues, but not in the serum samples. Moreover, there was a significant difference in the ratio of the active form (62 kDa) and proform (72 kDa) of MMP-2 among normal, BPH and prostate cancer tissues. This ratio was further increased in metastatic prostate cancer tissues.
Conclusion: The activity of MMP-9 and the ratio of active form/proform of MMP-2 are associated with the progression and metastasis of prostate cancer.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 4 print issues and online access
$259.00 per year
only $64.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
John A, Tuszynski G . The role of matrix metalloproteinases in tumor angiogenesis and tumor metastasis. Pathol Oncol Res 2001; 7: 14–23.
McCawley LJ, Matrisian LM . Matrix metalloproteinases: multifunctional contributors to tumor progression. Mol Med Today 2000; 6: 149–156.
Stetler-Stevenson WG, Aznavoorian S, Liotta LA . Tumor cell interactions with the extracellular matrix during invasion and metastasis. Annu Rev Cell Biol 1993; 9: 541–573.
Stetler-Stevenson WG, Hewitt R, Corcoran M . Matrix metalloproteinases and tumor invasion from correlation and causality to the clinic. Semin Cancer Biol 1996; 7: 147–154.
Moses MA et al. Increased incidence of matrix metalloproteinases in urine of cancer patients. Cancer Res 1998; 58: 1395–1399.
Gohji K et al. Serum matrix metalloproteinase-2 and its density in men with prostate cancer as a new predictor of disease extension. Int J Cancer 1998; 79: 96–101.
Stearns ME, Rhim J, Wang M . Interleukin 10 (IL-10) inhibition of primary human prostate cancer-induced angiogenesis: IL-10 stimulation of tissue inhibitor of metalloproteinase-1 and inhibition of matrix metalloproteinase (MMP)-2/MMP-9 secretion. Clin Cancer Res 1999; 5: 189–196.
Wood M et al. In situ hybridization studies of metalloproteinases 2 and 9 and TIMP-1 and TIMP-2 expression in human prostate cancer. Clin Exp Metast 1997; 15: 246–258.
Kuniyasu H et al. Relative expression of type IV collagenase, E-cadherin, and vascular endothelial growth factor/vascular permeability factor in prostatectomy specimens distinguishes organ-confined from pathologically advanced prostate cancers. Clin Cancer Res 2000; 6: 2295–2308.
Hiroki K et al. The relative mRNA expression levels of matrix metalloproteinases to E-cadherin in prostate biopsy specimens distinguishes organ-confined from advanced prostate cancer at radical prostatectomy. Clin Cancer Res 2003; 9: 2185–2194.
Strauss BH et al. In vivo collagen turnover following experimental balloon angioplasty injury and the role of matrix metalloproteinases. Circ Res 1996; 79: 541–550.
Zhang J et al. Differential expression of matrix metalloproteinases and their tissue inhibitors in human primary cultured prostatic cells and malignant cell lines. The Prostate 2002; 50: 38–45.
Sehgal I, Thompson TC . Novel regulation of type IV collagenase (matrix metalloproteinase-9 and -2) activities by transforming growth factor-β1 in human prostate cancer cell lines. Mol Biol Cell 1999; 10: 407–416.
Sato H, Seiki M . Regulatory mechanism of 92 kDa type IV collagenase gene expression which is associated with invasiveness of tumor cells. Oncogene 1993; 8: 395–405.
Benbow U, Brinckerhoff CE . The AP-1 site and MMP gene regulation: what is all the fuss about? Matrix Biol 1997; 15: 519–526.
Zhao YG et al. Activation of pro-gelatinase B by endometase/matrilysin-2 promotes invasion of human prostate cancer cells. J Biol Chem 2003; 278: 15056–15064.
Mazzieri R et al. Control of type IV collagenase activity by components of the urokinase–plasmin system: a regulatory mechanism with cell-bound reactants. EMBO J 1997; 16: 2319–2332.
Brown PD et al. Expression of activated gelatinase in human invasive breast carcinoma. Clin Exp Metast 1993; 11: 183–189.
Brown PD et al. Association between expression of activated 72-kD gelatin and tumor spread in non-small-cell lung carcinoma. J Natl Cancer Ins 1993; 85: 574–578.
Shiomi T, Okada Y . MT1-MMP and MMP-7 in invasion and metastasis of human cancers. Cancer Metast Rev 2003; 22: 145–152.
Garbisa S et al. Correlation of serum metalloproteinase levels with lung cancer metastasis and response to therapy. Cancer Res 1992; 52: 4548–4549.
Zucker S, Lysik RM, Zarrabi MH, Moll U . Mr 92,000 type IV collagenase is increased in plasma of patients with colon cancer and breast cancer. Cancer Res 1993; 53: 140–146.
Sauer CG et al. Expression and activity of matrix metalloproteinases-2 and -9 in serum, core needle biopsies and tissue specimens of prostate cancer patients. Virchows Arch 2004; 444: 518–526.
Acknowledgements
This research was funded by the following grants: 3 × 3 Canada China Biotechnology Seed Grant, the National Natural Science Foundation of China (Grant No. 30271297), and the United States Department of Health and Human Services, National Institutes of Health (NCI) Prostate Cancer SPORE (Grant No. P50 CA90386-01).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhang, L., Shi, J., Feng, J. et al. Type IV collagenase (matrix metalloproteinase-2 and -9) in prostate cancer. Prostate Cancer Prostatic Dis 7, 327–332 (2004). https://doi.org/10.1038/sj.pcan.4500750
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.pcan.4500750
Keywords
This article is cited by
-
SOST silencing promotes proliferation and invasion and reduces apoptosis of retinoblastoma cells by activating Wnt/β-catenin signaling pathway
Gene Therapy (2017)
-
Knockdown of WAVE3 impairs HGF induced migration and invasion of prostate cancer cells
Cancer Cell International (2015)
-
The activation of TLR7 regulates the expression of VEGF, TIMP1, MMP2, IL-6, and IL-15 in Hela cells
Molecular and Cellular Biochemistry (2014)
-
Flavokawain B, a kava chalcone, inhibits growth of human osteosarcoma cells through G2/M cell cycle arrest and apoptosis
Molecular Cancer (2013)
-
Matrix metalloproteinases in cancer: their value as diagnostic and prognostic markers and therapeutic targets
Tumor Biology (2013)