Abstract
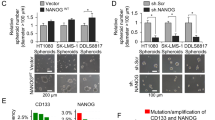
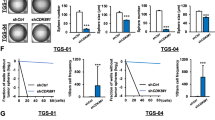
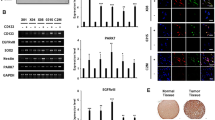
Uncovering the mechanisms that govern the maintenance of stem-like cancer cells is critical for developing therapeutic strategies for targeting these cells. Constitutive activation of c-Jun N-terminal kinase (JNK) has been reported in gliomas and correlates with histological grade. Here, we found that JNK signaling is crucial for the maintenance of ‘stemness’ in glioma cells. Sphere-cultured glioma cells showed more phosphorylation of JNK compared with serum-containing monolayer cultures. Importantly, blockade of JNK signaling with SP600125 or small interfering RNAs targeting JNK1 or JNK2 significantly reduced the CD133+/Nestin+ population and suppressed sphere formation, colony formation in soft agar, and expression of stem cell markers in sphere-cultured glioma cells. Intriguingly, sphere-cultured glioma cells exhibited enhanced expression of Notch-2, but not Notch-1, -3 or -4, and JNK inhibition almost completely abrogated this increase. Blocking the phosphoinoside 3-kinase (PI3K)/Akt pathway with LY294002 or si-Akt also suppressed the self-renewal of sphere-cultured glioma cells. PI3K, but not Akt, had a role as an upstream kinase in JNK1/2 activation. In addition, treatment with si-JNK greatly increased etoposide- and ionizing radiation (IR)-induced cell death in glioma spheres. Consistent with glioma cell lines, glioma stem-like cells isolated from primary patient glioma cells also had a higher activity of JNK and Notch-2 expression. Importantly, inhibition of JNK2 led to a decrease of Notch-2 expression and suppressed the CD133+/Nestin+ cell population in patient-derived primary glioma cells. Finally, downregulation of JNK2 almost completely suppressed intracranial tumor formation by glioma cells in nude mice. Taken together, these data demonstrate that JNK signaling is crucial for the maintenance of self-renewal and tumorigenicity of glioma stem-like cells and drug/IR resistance, and can be considered a promising target for eliminating stem-like cancer cells in gliomas.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Ohgaki H, Kleihues P . Population-based studies on incidence, survival rates, and genetic alterations in astrocytic and oligodendroglial gliomas. J Neuropathol Exp Neurol 2005; 64: 479–489.
Behin A, Hoang-Xuan K, Carpentier AF, Delattre JY . Primary brain tumours in adults. Lancet 2003; 361: 323–331.
Dirks PB . Brain tumor stem cells: bringing order to the chaos of brain cancer. J Clin Oncol 2008; 26: 2916–2924.
Lapidot T, Sirard C, Vormoor J, Murdoch B, Hoang T, Caceres-Cortes J et al. A cell initiating human acute myeloid leukaemia after transplantation into SCID mice. Nature 1994; 367: 645–648.
Singh SK, Hawkins C, Clarke ID, Squire JA, Bayani J, Hide T et al. Identification of human brain tumour initiating cells. Nature 2004; 432: 396–401.
Al-Hajj M, Wicha MS, Benito-Hernandez A, Morrison SJ, Clarke MF . Prospective identification of tumorigenic breast cancer cells. Proc Natl Acad Sci USA 2003; 100: 3983–3988.
Ricci-Vitiani L, Lombardi DG, Pilozzi E, Biffoni M, Todaro M, Peschle C et al. Identification and expansion of human colon-cancer-initiating cells. Nature 2007; 445: 111–115.
Eramo A, Lotti F, Sette G, Pilozzi E, Biffoni M, Di Virgilio A et al. Identification and expansion of the tumorigenic lung cancer stem cell population. Cell Death Differ 2008; 15: 504–514.
Visvader JE, Lindeman GJ . Cancer stem cells in solid tumours: accumulating evidence and unresolved questions. Nat Rev Cancer 2008; 8: 755–768.
Galli R, Binda E, Orfanelli U, Cipelletti B, Gritti A, De Vitis S et al. Isolation and characterization of tumorigenic, stem-like neural precursors from human glioblastoma. Cancer Res 2004; 64: 7011–7021.
Hemmati HD, Nakano I, Lazareff JA, Masterman-Smith M, Geschwind DH, Bronner-Fraser M et al. Cancerous stem cells can arise from pediatric brain tumors. Proc Natl Acad Sci USA 2003; 100: 15178–15183.
Salmaggi A, Boiardi A, Gelati M, Russo A, Calatozzolo C, Ciusani E et al. Glioblastoma-derived tumorospheres identify a population of tumor stem-like cells with angiogenic potential and enhanced multidrug resistance phenotype. Glia 2006; 54: 850–860.
Yi L, Zhou ZH, Ping YF, Chen JH, Yao XH, Feng H et al. Isolation and characterization of stem cell-like precursor cells from primary human anaplastic oligoastrocytoma. Mod Pathol 2007; 20: 1061–1068.
Murat A, Migliavacca E, Gorlia T, Lambiv WL, Shay T, Hamou MF et al. Stem cell-related ″self-renewal″ signature and high epidermal growth factor receptor expression associated with resistance to concomitant chemoradiotherapy in glioblastoma. J Clin Oncol 2008; 26: 3015–3024.
Bao S, Wu Q, McLendon RE, Hao Y, Shi Q, Hjelmeland AB et al. Glioma stem cells promote radioresistance by preferential activation of the DNA damage response. Nature 2006a; 444: 756–760.
Bao S, Wu Q, Sathornsumetee S, Hao Y, Li Z, Hjelmeland AB et al. Stem cell-like glioma cells promote tumor angiogenesis through vascular endothelial growth factor. Cancer Res 2006b; 66: 7843–7848.
Li Z, Bao S, Wu Q, Wang H, Eyler C, Sathornsumetee S et al. Hypoxia-inducible factors regulate tumorigenic capacity of glioma stem cells. Cancer Cell 2009; 15: 501–513.
Kennedy NJ, Davis RJ . Role of JNK in tumor development. Cell Cycle 2003; 2: 199–201.
Davis RJ . Signal transduction by the JNK group of MAP kinases. Cell 2000; 103: 239–252.
Tsuiki H, Tnani M, Okamoto I, Kenyon LC, Emlet DR, Holgado-Madruga M et al. Constitutively active forms of c-Jun NH2-terminal kinase are expressed in primary glial tumors. Cancer Res 2003; 63: 250–255.
Li JY, Wang H, May S, Song X, Fueyo J, Fuller GN . Constitutive activation of c-Jun N-terminal kinase correlates with histologic grade and EGFR expression in diffuse gliomas. J Neurooncol 2008; 88: 11–17.
Antonyak MA, Kenyon LC, Godwin AK, James DC, Emlet DR, Okamoto I et al. Elevated JNK activation contributes to the pathogenesis of human brain tumors. Oncogene 2002; 21: 5038–5046.
Cui J, Han SY, Wang C, Su W, Harshyne L, Holgado-Madruga M et al. c-Jun NH(2)-terminal kinase 2alpha2 promotes the tumorigenicity of human glioblastoma cells. Cancer Res 2006; 66: 10024–10031.
Joseph NM, Morrison SJ . Toward an understanding of the physiological function of Mammalian stem cells. Dev Cell 2005; 9: 173–183.
Chiba S . Notch signaling in stem cell systems. Stem Cells 2006; 24: 2437–2447.
Wang Z, Li Y, Banerjee S, Sarkar FH . Emerging role of Notch in stem cells and cancer. Cancer Lett 2009; 279: 8–12.
Somasundaram K, Reddy SP, Vinnakota K, Britto R, Subbarayan M, Nambiar S et al. Upregulation of ASCL1 and inhibition of Notch signaling pathway characterize progressive astrocytoma. Oncogene 2005; 24: 7073–7083.
Chen J, Xu J, Zhao W, Hu G, Cheng H, Kang Y et al. Characterization of human LNX, a novel ligand of Numb protein X that is downregulated in human gliomas. Int J Biochem Cell Biol 2005; 37: 2273–2283.
Zhang XP, Zheng G, Zou L, Liu HL, Hou LH, Zhou P et al. Notch activation promotes cell proliferation and the formation of neural stem cell-like colonies in human glioma cells. Mol Cell Biochem 2008; 307: 101–108.
Wang J, Wakeman TP, Lathia JD, Hjelmeland AB, Wang XF, White RR et al. Notch promotes radioresistance of glioma stem cells. Stem Cells 2010; 28: 17–28.
Purow BW, Haque RM, Noel MW, Su Q, Burdick MJ, Lee J et al. Expression of Notch-1 and its ligands, Delta-like-1 and Jagged-1, is critical for glioma cell survival and proliferation. Cancer Res 2005; 65: 2353–2363.
Kanamori M, Kawaguchi T, Nigro JM, Feuerstein BG, Berger MS, Miele L et al. Contribution of Notch signaling activation to human glioblastoma multiforme. J Neurosurg 2007; 106: 417–427.
Fan X, Khaki L, Zhu TS, Soules ME, Talsma CE, Gul N et al. NOTCH pathway blockade depletes CD133-positive glioblastoma cells and inhibits growth of tumor neurospheres and xenografts. Stem Cells 2010; 28: 5–16.
Zhong Y, Zhou C, Ma W, Wang D, Guo S, Su X et al. Most MCF7 and SK-OV3 cells were deprived of their stem nature by Hoechst 33342. Biochem Biophys Res Commun 2007; 364: 338–343.
Stiles CD, Rowitch DH . Glioma stem cells: a midterm exam. Neuron 2008; 58: 832–846.
Yu SC, Ping YF, Yi L, Zhou ZH, Chen JH, Yao XH et al. Isolation and characterization of cancer stem cells from a human glioblastoma cell line U87. Cancer Lett 2008; 265: 124–134.
Bleau AM, Hambardzumyan D, Ozawa T, Fomchenko EI, Huse JT, Brennan CW et al. PTEN/PI3K/Akt pathway regulates the side population phenotype and ABCG2 activity in glioma tumor stem-like cells. Cell Stem Cell 2009; 4: 226–235.
Zhang M, Atkinson RL, Rosen JM . Selective targeting of radiation-resistant tumor-initiating cells. Proc Natl Acad Sci USA 2010; 107: 3522–3527.
Frosina G . DNA repair and resistance of gliomas to chemotherapy and radiotherapy. Mol Cancer Res 2009; 7: 989–999.
Soeda A, Park M, Lee D, Mintz A, Androutsellis-Theotokis A, McKay RD et al. Hypoxia promotes expansion of the CD133-positive glioma stem cells through activation of HIF-1alpha. Oncogene 2009; 28: 3949–3959.
Zhou J, Wulfkuhle J, Zhang H, Gu P, Yang Y, Deng J et al. Activation of the PTEN/mTOR/STAT3 pathway in breast cancer stem-like cells is required for viability and maintenance. Proc Natl Acad Sci USA 2007; 104: 16158–16163.
Dubrovska A, Kim S, Salamone RJ, Walker JR, Maira SM, Garcia-Echeverria C et al. The role of PTEN/Akt/PI3K signaling in the maintenance and viability of prostate cancer stem-like cell populations. Proc Natl Acad Sci USA 2009; 106: 268–273.
Hambardzumyan D, Becher OJ, Rosenblum MK, Pandolfi PP, Manova-Todorova K, Holland EC . PI3K pathway regulates survival of cancer stem cells residing in the perivascular niche following radiation in medulloblastoma in vivo. Genes Dev 2008; 22: 436–448.
Kanzawa T, Iwado E, Aoki H, Iwamaru A, Hollingsworth EF, Sawaya R et al. Ionizing radiation induces apoptosis and inhibits neuronal differentiation in rat neural stem cells via the c-Jun NH2-terminal kinase (JNK) pathway. Oncogene 2006; 25: 3638–3648.
Potapova O, Gorospe M, Bost F, Dean NM, Gaarde WA, Mercola D et al. c-Jun N-terminal kinase is essential for growth of human T98G glioblastoma cells. J Biol Chem 2000; 275: 24767–24775.
Mangiola A, Lama G, Giannitelli C, De Bonis P, Anile C, Lauriola L et al. Stem cell marker nestin and c-Jun NH2-terminal kinases in tumor and peritumor areas of glioblastoma multiforme: possible prognostic implications. Clin Cancer Res 2007; 13: 6970–6977.
Xu P, Qiu M, Zhang Z, Kang C, Jiang R, Jia Z et al. The oncogenic roles of Notch1 in astrocytic gliomas in vitro and in vivo. J Neurooncol 2010; 97: 41–51.
Fan X, Matsui W, Khaki L, Stearns D, Chun J, Li YM et al. Notch pathway inhibition depletes stem-like cells and blocks engraftment in embryonal brain tumors. Cancer Res 2006; 66: 7445–7452.
Shih AH, Holland EC . Notch signaling enhances nestin expression in gliomas. Neoplasia 2006; 8: 1072–1082.
Gaiano N, Fishell G . The role of notch in promoting glial and neural stem cell fates. Annu Rev Neurosci 2002; 25: 471–490.
Yoon K, Gaiano N . Notch signaling in the mammalian central nervous system: insights from mouse mutants. Nat Neurosci 2005; 8: 709–715.
Mizutani K, Yoon K, Dang L, Tokunaga A, Gaiano N . Differential Notch signalling distinguishes neural stem cells from intermediate progenitors. Nature 2007; 449: 351–355.
Hovinga KE, Shimizu F, Wang R, Panagiotakos G, Van Der Heijden M, Moayedpardazi H et al. Inhibition of notch signaling in glioblastoma targets cancer stem cells via an endothelial cell intermediate. Stem Cells 2010; 28: 1019–1029.
Eyler CE, Wu Q, Yan K, MacSwords JM, Chandler-Militello D, Misuraca KL et al. Glioma stem cell proliferation and tumor growth are promoted by nitric oxide synthase-2. Cell 2011; 146: 53–66.
Charles N, Ozawa T, Squatrito M, Bleau AM, Brennan CW, Hambardzumyan D et al. Perivascular nitric oxide activates notch signaling and promotes stem-like character in PDGF-induced glioma cells. Cell Stem Cell 2010; 6: 141–152.
Mishra OP, Zubrow AB, Ashraf QM . Nitric oxide-mediated activation of extracellular signal-regulated kinase (ERK) and c-jun N-terminal kinase (JNK) during hypoxia in cerebral cortical nuclei of newborn piglets. Neuroscience 2004; 123: 179–186.
Soeda A, Inagaki A, Oka N, Ikegame Y, Aoki H, Yoshimura S et al. Epidermal growth factor plays a crucial role in mitogenic regulation of human brain tumor stem cells. J Biol Chem 2008; 283: 10958–10966.
Suslov ON, Kukekov VG, Ignatova TN, Steindler DA . Neural stem cell heterogeneity demonstrated by molecular phenotyping of clonal neurospheres. Proc Natl Acad Sci USA 2002; 99: 14506–14511.
Singh SK, Clarke ID, Terasaki M, Bonn VE, Hawkins C, Squire J et al. Identification of a cancer stem cell in human brain tumors. Cancer Res 2003; 63: 5821–5828.
Singh A, Greninger P, Rhodes D, Koopman L, Violette S, Bardeesy N et al. A gene expression signature associated with ″K-Ras addiction″ reveals regulators of EMT and tumor cell survival. Cancer Cell 2009; 15: 489–500.
Moffat J, Grueneberg DA, Yang X, Kim SY, Kloepfer AM, Hinkle G et al. A lentiviral RNAi library for human and mouse genes applied to an arrayed viral high-content screen. Cell 2006; 124: 1283–1298.
Hyun KH, Yoon CH, Kim RK, Lim EJ, An S, Park MJ et al. Eckol suppresses maintenance of stemness and malignancies in glioma stem-like cells. Toxicol Appl Pharmacol 2011; 254: 32–40.
Acknowledgements
We thank Dr Akio Soeda (Department of Neurological Surgery, University of Virginia, USA) for providing patient-derived glioma cells. This work was supported by the Korea Research Foundation (KRF) and Ministry of Education, Science and Technology (MEST), Korean government, through its National Nuclear Technology Program (2008-2003935) and the National R&D Program for Cancer Control, Ministry for Health, Welfare and Family affairs, Republic of Korea (1020340).
Author contributions: Chang-Hwan Yoon and Min-Jung Kim helped in the collection and assembly of data, data analysis and interpretation. Rae-Kwon Kim and Eun-Jung Lim helped in collection of data. Kyu-Sil Choi, Sungkwan An, Sang-Gu Hwang and Kang Seok-Gu helped in data analysis and interpretation. Yongjoon Suh helped in data analysis and interpretation, manuscript writing. Myung-Jin Parkhelped in manuscript writing, data analysis and interpretation, provision of study material or patients. Su-Jae Lee helped in conception and design, manuscript writing, data analysis and interpretation, final approval of manuscript.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Oncogene website
Supplementary information
Rights and permissions
About this article
Cite this article
Yoon, CH., Kim, MJ., Kim, RK. et al. c-Jun N-terminal kinase has a pivotal role in the maintenance of self-renewal and tumorigenicity in glioma stem-like cells. Oncogene 31, 4655–4666 (2012). https://doi.org/10.1038/onc.2011.634
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2011.634
Keywords
This article is cited by
-
Current and future therapeutic strategies for high-grade gliomas leveraging the interplay between epigenetic regulators and kinase signaling networks
Journal of Experimental & Clinical Cancer Research (2024)
-
Kunitz-type protease inhibitor TFPI2 remodels stemness and immunosuppressive tumor microenvironment in glioblastoma
Nature Immunology (2023)
-
Pharmacogenomic profiling reveals molecular features of chemotherapy resistance in IDH wild-type primary glioblastoma
Genome Medicine (2023)
-
Depleting TMED3 alleviates the development of endometrial carcinoma
Cancer Cell International (2022)
-
Selective antagonism of cJun for cancer therapy
Journal of Experimental & Clinical Cancer Research (2020)