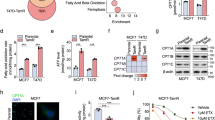
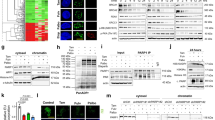
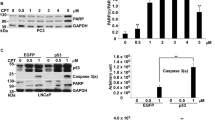
Abstract
The c-Jun N-terminal kinase (JNK) has been shown to mediate tamoxifen-induced apoptosis in breast cancer cells. However, the downstream mediators of the JNK pathway linking tamoxifen to effectors of apoptosis have yet to be identified. In this study, we analysed whether c-Jun, the major nuclear target of JNK, has a role in tamoxifen-induced apoptosis of SkBr3 breast cancer cells. We show that before DNA fragmentation and caspase 3/7 activation, cytotoxic concentrations of 4-hydroxytamoxifen (OHT) induced JNK-dependent phosphorylation of c-Jun at JNK sites earlier shown to regulate c-Jun-mediated apoptosis. In addition, OHT induced ERK-dependent expression of c-Fos and transactivation of an AP-1-responsive promoter. In particular, the ectopic expression of dominant-negative constructs blocking either AP-1 activity or c-Jun N-terminal phosphorylation prevented DNA fragmentation after OHT treatment. Furthermore, both c-Fos expression and c-Jun N-terminal phosphorylation preceded OHT-dependent activation of caspase 3–7 in different types of tamoxifen-sensitive cancer cells, but not in OHT-resistant LNCaP prostate cancer cells. Taken together, our results indicate that the c-Jun/c-Fos AP-1 complex has a pro-apoptotic role in OHT-treated cancer cells and suggest that pharmacological boosts of c-Jun activation may be useful in a combination therapy setting to sensitize cancer cells to tamoxifen-mediated cell death.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Albanito L, Madeo A, Lappano R, Vivacqua A, Rago V, Carpino A et al. (2007). G protein-coupled receptor 30 (GPR30) mediates gene expression changes and growth response to 17beta-estradiol and selective GPR30 ligand G-1 in ovarian cancer cells. Cancer Res 67: 1859–1866.
Albanito L, Sisci D, Aquila S, Brunelli E, Vivacqua A, Madeo A et al. (2008). Epidermal growth factor induces G protein-coupled receptor 30 expression in estrogen receptor-negative breast cancer cells. Endocrinology 149: 3799–3808.
Ballatori N, Krance SM, Notenboom S, Shi S, Tieu K, Hammond CL . (2009). Glutathione dysregulation and the etiology and progression of human diseases. Biol Chem 390: 191–214.
Bannister AJ, Oehler T, Wilhelm D, Angel P, Kouzarides T . (1995). Stimulation of c-Jun activity by CBP: c-Jun residues Ser63/73 are required for CBP induced stimulation in vivo and CBP binding in vitro. Oncogene 11: 2509–2514.
Basu S, Ma R, Boyle PJ, Mikulla B, Bradley M, Smith B et al. (2004). Apoptosis of human carcinoma cells in the presence of potential anti-cancer drugs: III. Treatment of Colo-205 and SKBR3 cells with: cis -platin, tamoxifen, melphalan, betulinic acid, L-PDMP, L-PPMP, and GD3 ganglioside. Glycoconj J 20: 563–577.
Behrens A, Sibilia M, Wagner EF (1999). Amino-terminal phosphorylation of c-Jun regulates stress-induced apoptosis and cellular proliferation. Nat Genet 21: 326–329.
Davis RJ . (2000). Signal transduction by the JNK group of MAP kinases. Cell 103: 239–252.
Dunn C, Wiltshire C, MacLaren A, Gillespie DA . (2002). Molecular mechanism and biological functions of c-Jun N-terminal kinase signalling via the c-Jun transcription factor. Cell Signal 14: 585–593.
Eferl R, Wagner EF . (2003). AP-1: a double-edged sword in tumorigenesis. Nat Rev Cancer 3: 859–868.
Friesen C, Kiess Y, Debatin KM . (2004). A critical role of glutathione in determining apoptosis sensitivity and resistance in leukemia cells. Cell Death Differ 11 (Suppl 1): S73–S85.
Ham J, Towers E, Gilley J, Terzano S, Randall R . (2005). BH3-only proteins: key regulators of neuronal apoptosis. Cell Death Differ 12: 1015–1020.
Hess J, Angel P, Schorpp-Kistner M . (2004). AP-1 subunits: quarrel and harmony among siblings. J Cell Sci 117: 5965–5973.
Kallio A, Zheng A, Dahllund J, Heiskanen KM, Harkonen P . (2005). Role of mitochondria in tamoxifen-induced rapid death of MCF-7 breast cancer cells. Apoptosis 10: 1395–1410.
Lagadec C, Adriaenssens E, Toillon RA, Chopin V, Romon R, Van Coppenolle F et al. (2008). Tamoxifen and TRAIL synergistically induce apoptosis in breast cancer cells. Oncogene 27: 1472–1477.
Lee YS, Kang YS, Lee SH, Kim JA . (2000). Role of NAD(P)H oxidase in the tamoxifen-induced generation of reactive oxygen species and apoptosis in HepG2 human hepatoblastoma cells. Cell Death Differ 7: 925–932.
Liu H, Sidiropoulos P, Song G, Pagliari LJ, Birrer MJ, Stein B et al. (2000). TNF-alpha gene expression in macrophages: regulation by NF-kappa B is independent of c-Jun or C/EBP beta. J Immunol 164: 4277–4285.
Love RR . (1989). Tamoxifen therapy in primary breast cancer: biology, efficacy, and side effects. J Clin Oncol 7: 803–815.
Mabuchi S, Ohmichi M, Kimura A, Ikebuchi Y, Hisamoto K, Arimoto-Ishida E et al. (2004). Tamoxifen inhibits cell proliferation via mitogen-activated protein kinase cascades in human ovarian cancer cell lines in a manner not dependent on the expression of estrogen receptor or the sensitivity to cisplatin. Endocrinology 145: 1302–1313.
Maggiolini M, Vivacqua A, Fasanella G, Recchia AG, Sisci D, Pezzi V et al. (2004). The G protein-coupled receptor GPR30 mediates c-fos up-regulation by 17beta-estradiol and phytoestrogens in breast cancer cells. J Biol Chem 279: 27008–27016.
Mandlekar S, Kong AN . (2001). Mechanisms of tamoxifen-induced apoptosis. Apoptosis 6: 469–477.
Mandlekar S, Yu R, Tan TH, Kong AN . (2000). Activation of caspase-3 and c-Jun NH2-terminal kinase-1 signaling pathways in tamoxifen-induced apoptosis of human breast cancer cells. Cancer Res 60: 5995–6000.
Morton S, Davis RJ, McLaren A, Cohen P . (2003). A reinvestigation of the multisite phosphorylation of the transcription factor c-Jun. Embo J 22: 3876–3886.
Musti AM, Treier M, Bohmann D . (1997). Reduced ubiquitin-dependent degradation of c-Jun after phosphorylation by MAP kinases. Science 275: 400–402.
Nateri AS, Spencer-Dene B, Behrens A . (2005). Interaction of phosphorylated c-Jun with TCF4 regulates intestinal cancer development. Nature 437: 281–285.
Nephew KP, Polek TC, Akcali KC, Khan SA . (1993). The antiestrogen tamoxifen induces c-fos and jun-B, but not c-jun or jun-D, protooncogenes in the rat uterus. Endocrinology 133: 419–422.
Olive M, Krylov D, Echlin DR, Gardner K, Taparowsky E, Vinson C . (1997). A dominant negative to activation protein-1 (AP1) that abolishes DNA binding and inhibits oncogenesis. J Biol Chem 272: 18586–18594.
Papavassiliou AG, Treier M, Bohmann D . (1995). Intramolecular signal transduction in c-Jun. Embo J 14: 2014–2019.
Papavassiliou AG, Treier M, Chavrier C, Bohmann D . (1992). Targeted degradation of c-Fos, but not v-Fos, by a phosphorylation-dependent signal on c-Jun. Science 258: 1941–1944.
Pope R, Mungre S, Liu H, Thimmapaya B . (2000). Regulation of TNF-alpha expression in normal macrophages: the role of C/EBPbeta. Cytokine 12: 1171–1181.
Porter AG, Janicke RU . (1999). Emerging roles of caspase-3 in apoptosis. Cell Death Differ 6: 99–104.
Price MA, Rogers AE, Treisman R . (1995). Comparative analysis of the ternary complex factors Elk-1, SAP-1a and SAP-2 (ERP/NET). Embo J 14: 2589–2601.
Prossnitz ER, Maggiolini M . (2009). Mechanisms of estrogen signaling and gene expression via GPR30. Mol Cell Endocrinol 308: 32–38.
Raivich G . (2008). c-Jun expression, activation and function in neural cell death, inflammation and repair. J Neurochem 107: 898–906.
Raivich G, Behrens A . (2006). Role of the AP-1 transcription factor c-Jun in developing, adult and injured brain. Prog Neurobiol 78: 347–363.
Recktenwald CV, Kellner R, Lichtenfels R, Seliger B . (2008). Altered detoxification status and increased resistance to oxidative stress by K-ras transformation. Cancer Res 68: 10086–10093.
Schiff R, Reddy P, Ahotupa M, Coronado-Heinsohn E, Grim M, Hilsenbeck SG et al. (2000). Oxidative stress and AP-1 activity in tamoxifen-resistant breast tumors in vivo. J Natl Cancer Inst 92: 1926–1934.
Shaulian E, Karin M . (2001). AP-1 in cell proliferation and survival. Oncogene 20: 2390–2400.
Shen HM, Shi CY, Shen Y, Ong CN . (1996). Detection of elevated reactive oxygen species level in cultured rat hepatocytes treated with aflatoxin B1. Free Radic Biol Med 21: 139–146.
Smith CL, Nawaz Z, O’Malley BW . (1997). Coactivator and corepressor regulation of the agonist/antagonist activity of the mixed antiestrogen, 4-hydroxytamoxifen. Mol Endocrinol 11: 657–666.
Strober W . (2001). Trypan blue exclusion test of cell viability. Curr Protoc Immunol; Appendix 3: A.3B.1-2, Supplement 21.
Treisman R . (1995). Journey to the surface of the cell: Fos regulation and the SRE. Embo J 14: 4905–4913.
Vinciguerra M, Esposito I, Salzano S, Madeo A, Nagel G, Maggiolini M et al. (2008). Negative charged threonine 95 of c-Jun is essential for c-Jun N-terminal kinase-dependent phosphorylation of threonine 91/93 and stress-induced c-Jun biological activity. Int J Biochem Cell Biol 40: 307–316.
Vivacqua A, Bonofiglio D, Albanito L, Madeo A, Rago V, Carpino A et al. (2006a). 17beta-estradiol, genistein, and 4-hydroxytamoxifen induce the proliferation of thyroid cancer cells through the g protein-coupled receptor GPR30. Mol Pharmacol 70: 1414–1423.
Vivacqua A, Bonofiglio D, Recchia AG, Musti AM, Picard D, Ando S et al. (2006b). The G protein-coupled receptor GPR30 mediates the proliferative effects induced by 17beta-estradiol and hydroxytamoxifen in endometrial cancer cells. Mol Endocrinol 20: 631–646.
Wada T, Joza N, Cheng HY, Sasaki T, Kozieradzki I, Bachmaier K et al. (2004). MKK7 couples stress signalling to G2/M cell-cycle progression and cellular senescence. Nat Cell Biol 6: 215–226.
Watson A, Eilers A, Lallemand D, Kyriakis J, Rubin LL, Ham J . (1998). Phosphorylation of c-Jun is necessary for apoptosis induced by survival signal withdrawal in cerebellar granule neurons. J Neurosci 18: 751–762.
Weiss C, Schneider S, Wagner EF, Zhang X, Seto E, Bohmann D . (2003). JNK phosphorylation relieves HDAC3-dependent suppression of the transcriptional activity of c-Jun. Embo J 22: 3686–3695.
Whitfield J, Neame SJ, Paquet L, Bernard O, Ham J . (2001). Dominant-negative c-Jun promotes neuronal survival by reducing BIM expression and inhibiting mitochondrial cytochrome c release. Neuron 29: 629–643.
Young TW, Mei FC, Yang G, Thompson-Lanza JA, Liu J, Cheng X . (2004). Activation of antioxidant pathways in ras-mediated oncogenic transformation of human surface ovarian epithelial cells revealed by functional proteomics and mass spectrometry. Cancer Res 64: 4577–4584.
Acknowledgements
We thank Dr Lidia Albanito for continuous helpful experimental assistance. We thank Dr Anna Grazia Recchia for the English revision of the paper. We are grateful to C Vinson, K Nose and H van Dam for gifts of reagents. Funding for this project was provided by the Associazione Italiana Ricerca sul Cancro (AIRC), Ministero dell’Universita' e Ricerca, Regione Calabria.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Supplementary Information accompanies the paper on the Oncogene website (http://www.nature.com/onc)
Rights and permissions
About this article
Cite this article
Madeo, A., Vinciguerra, M., Lappano, R. et al. c-Jun activation is required for 4-hydroxytamoxifen-induced cell death in breast cancer cells. Oncogene 29, 978–991 (2010). https://doi.org/10.1038/onc.2009.400
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2009.400