Abstract
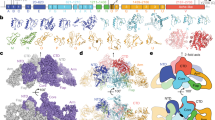
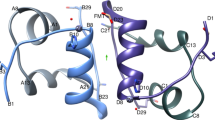
Since its first isolation1, bovine β-lactoglobulin (BLG) has been an enigma: although it is abundant in the whey fraction of milk, its function is still not clear. The results of the many physicochemical studies on the protein need a structural interpretation. We report here the structure of the orthorhombic crystal form of cow BLG at pH 7.6, at a resolution of 2.8 Å. It has an unusual protein fold, composed of two slabs of antiparallel β-sheet, which shows a remarkable similarity to plasma retinol-binding protein. A possible binding site for retinol in BLG has been identified by model-building. This suggests a role for BLG in vitamin A transport and we have discovered specific receptors for the BLG–retinol complex in the intestine of neonate calves.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Palmer, A. H. J. biol. Chem. 104, 359–372 (1934).
Pervaiz, S. & Brew, K. Science 228, 335–337 (1985).
Grosclaude, F., Mahe, M. F., Mercier, J. C., Bonnmaire, J. & Teissier, J. H. Ann. Genet. Sel. anim. 8, 461–479 (1976).
McKenzie, H. A. in Milk Proteins: Chemistry and Molecular Biology Vol. 2 (ed. McKenzie, H. A.) 257–330 (Academic, New York, 1971).
Martial, J., Preaux, G. & Lontie, R. Arch. int. Physiol. Biochim. 79, 842–843 (1971).
Mainferme, F., Preaux, G. & Lontie, R. Arch. int. Physiol. Biochim. 79, 840–841 (1971).
McKenzie, H. A., Ralton, G. B. & Shaw, D. C. Biochemistry 11, 4539–4547 (1972).
Lyster, R. L. J. J. Diary Res. 39, 279–318 (1972).
Jenness, R. in Lactation: a Comprehensive Treatise, Vol. 3, (eds Larson, B. L. & Smith, V. R.) 3–107 (Academic, New York, 1974).
Jenness, R. J. Diary Res. 46, 197–210 (1979).
Aschaffenburg, R. & Drewry, J. Biochem. J. 65, 273–277 (1957).
Miranda, G. & Pelissier, J. P. J. Dairy Res. 50, 27–36 (1983).
Tanford, C., Bunville, L. G. & Nozaki, Y. J. Am. chem. Soc. 81, 4032–4036 (1959).
Lovrien, R. & Andersen, W. F. Arch. Biochem. Biophys. 131, 139–144 (1969).
Futterman, S. & Heller, J. J. biol. Chem. 247, 5168–5172 (1972).
Stone, W. L. & Wishnia, A. Bioinorg. Chem. 8, 517–529 (1979).
Aschaffenburg, R., Green, D. W. & Simmons, R. M. J. molec. Biol. 13, 194–210 (1965).
Bolognesi, M., Liberatori, J., Oberti, R. & Ungaretti, L. J. molec. Biol. 131, 411–413 (1979).
Green, D. W. et al. J. molec. Biol. 131, 375–397 (1979).
Sawyer, L., Papiz, M. Z., North, A. C. T. & Eliopoulos, E. E. Biochem. Soc. Trans. 13, 265–266 (1985).
Rask, L. et al. Scand. J. clin. Lab. Invest. 40, Suppl. 154, 45–61 (1980).
Goodman, D. S. The Retinoids Vol. 2, 41–48 (1984).
Newcomer, M. E. et al. EMBO J. 3, 1451–1454 (1984).
Jones, T. A. & Newcomer, M. E. J. molec. Biol. (submitted).
Murthy, M. R. N., Reid, T. J., Sicgnano, A., Tanaka, N. & Rossmann, M. G. J. molec. Biol. 152, 465–499 (1981).
Chothia, C. & Janin, J. Biochemistry 21, 3955–3965 (1982).
Godovac-Zimmermann, J., Conti, A., Liberatori, J. & Braunitzer, G. Hoppe-Seyler Z. Phys. Chem. 366, 431–434 (1985).
Devereux, J., Haeberli, P. & Smithies, O. Nucleic Acid Res. 12, 387–395 (1984).
Rao, S. T. & Rossmann, M. G. J. molec. Biol. 76, 241–256 (1973).
Rossmann, M. G. & Argos, P. J. biol. Chem. 250, 7525–7537 (1975).
Fugate, F. D. & Song, P.-S., Biochim. biophys. Acta 652, 28–42 (1980).
Horwitz, J. & Heller, J. J. biol. Chem. 249, 4712–4719 (1974).
Cogan, U., Kopelman, M., Mokady, S. & Shinitzky, M. Eur. J. Biochem. 65, 71–78 (1976).
Cambillau, C., Horjales, E. & Jones, T. A. J. mol Graph. 2, 53–54 (1984).
Jones, T. A. J. appl. Cryst. 11, 268–272 (1978).
Eklund, H. et al. J. molec. Biol. 146, 561–587 (1981).
Hemley, R., Kohler, B. E. & Siviski, P. Biophys. J. 28, 447–455 (1979).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Papiz, M., Sawyer, L., Eliopoulos, E. et al. The structure of β-lactoglobulin and its similarity to plasma retinol-binding protein. Nature 324, 383–385 (1986). https://doi.org/10.1038/324383a0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/324383a0
This article is cited by
-
Reduced immunogenicity of β-lactoglobulin by single amino acid substitution
Cytotechnology (2022)
-
Binding analysis of the curcumin-based synthetic alpha-glucosidase inhibitors to beta-lactoglobulin as potential vehicle carrier for antidiabetic drugs
Journal of the Iranian Chemical Society (2022)
-
Effect of composition and drying method on glass transition temperature, water sorption characteristics and surface morphology of newly designed β-lactoglobulin/retinyl palmitate/disaccharides systems
Journal of Thermal Analysis and Calorimetry (2017)
-
Current ways to modify the structure of whey proteins for specific functionalities—a review
Dairy Science & Technology (2015)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.