Abstract
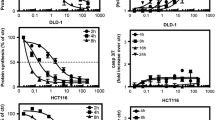
Immunotoxins are presently being evaluated as novel agents for cancer therapy. The direct mechanism by which immunotoxins kill cancer cells is inhibition of protein synthesis, but cytotoxicity due to induction of apoptosis has also been observed with these agents. Some cancers that express high levels of BCL-2 are relatively resistant to apoptosis inducing agents. It is therefore important to determine to what degree the toxicity of ricin, diphtheria toxin, Pseudomonas exotoxin and Pseudomonas exotoxin derived immunotoxins towards cancer cells can be attributed to inhibition of protein synthesis, and to what degree to subsequent induction of apoptosis. We compared the sensitivity of MCF-7 breast cancer cells that were stably transfected with a BCL-2 expression plasmid and thus protected against apoptosis and of MCF-7 cells transfected with a control plasmid towards ricin, diphtheria and Pseudomonas toxin, a Pseudomonas toxin-derived immunotoxin (LMB-7) and tumour necrosis factor α (TNF). We found that BCL-2 mediated inhibition of apoptosis renders the cells almost completely resistant (1000-fold) to tumour necrosis factor, but the same cells were only 3–10 fold more resistant to cytotoxicity induced by immunotoxin LMB-7 as well as Pseudo-monas exotoxin, diphtheria toxin and ricin. We next studied several leukaemia cell lines with variable levels of BCL-2 expression and found them quite sensitive to a Pseudomonas exotoxin containing immunotoxin independent of the level of BCL-2. Our data indicate that although BCL-2 overexpression can have a modest effect on sensitivity to an immunotoxin, cell lines derived from patients are still very sensitive to immunotoxins.
Similar content being viewed by others
References
Vitetta ES, Thorpe PE, Uhr JW. Immunotoxins: magic bullets or misguided missiles? Trends Pharmaco Sci 1993; 14: 148–154.
Pastan I, Pai LH, Brinkmann U, FitzGerald DJ. Recombinant toxins: new therapeutic agents for cancer. Ann NY Acad Sci 1995; 758: 345–354.
Brinkmann U, Pastan I. Immunotoxins against cancer. Biochem Biophys Acta 1994; 1198: 27–45.
Kreitman RJ, Pastan I. Recombinant single-chain immunotoxins against T and B cell leukemias. Leuk Lymphoma 1994; 13: 1–10.
Kochi SK, Collier RJ. DNA fragmentation and cytolysis in U937 cells treated with diphtheria toxin or other inhibitors of protein synthesis. Exp Cell Res 1993; 208: 296–302.
Chang MP, Bramhall J, Graves S, Bonavida B, Wisniesky BJ. Internucleosomal DNA cleavage preceded diphtheria toxin-induced cytolysis. Evidence that cell lysis is not a simple consequence of translation inhibition. J Biol Chem 1989; 264: 15261–15267.
Morimoto H, Bonavida B. Diphtheria toxin-and Pseudomonas A toxin-mediated apoptosis. ADP ribosylation of elongation factor-2 is required for DNA fragmentation and cell lysis and synergy with tumour necrosis factor-alpha. J Immunol 1992; 149: 2089–2094.
Tsujimoto Y, Cossman J, Jaffe E, Croce CM. Involvement of the BCL-2 gene in human follicular lymphoma. Science 1985; 228: 1440–1443.
Sentman CL, Shutter JR, Hockenbery D, Kanagawa O, Korsmeyer SJ. Bcl-2 inhibits multiple forms of apoptosis but not negative selection in thymocytes. Cell 1991; 67: 879–888.
Brinkmann U, Brinkmann E, Pastan I. Immunotoxin selection cloning of cDNAs that render cancer cells resistant to Pseudomonas and Diphtheria toxin and immunotoxins. Mol Med 1995; 1: 206–216.
Brinkmann U, Pai LH, FitzGerald DJ, Willingham M, Pastan I. B3(Fv)-PE38KDEL, a single-chain immunotoxin that causes complete regression of a human carcinoma in mice. Proc Natl Acad Sci USA 1991; 88: 8616–20.
Jinno Y, Chaudhary VK, Willingham MC, Adhya S, Pastan I. Domain II mutants of Pseudomonas exotoxin deficient in translocation. J Biol Chem 1988; 264: 15953–15959.
Buchner J, Pastan I, Brinkmann U. A method to increase the yield of properly folded recombinant fusion proteins: single-chain immunotoxins from renaturation of bacterial inclusion bodies. Anal Biochem 1992; 205: 263–270.
Brinkmann U, Pastan I. Recombinant immunotoxins: from basic research to cancer therapy. Methods 1995; 8: 143–156.
Li J-L, Shen G-L, Ghetie M-A, May RD, Till M, Ghetie V, et al. The epitope specificity and tissue reactivity of four murine monoclonal anti-CD22 antibodies. Cell Immunol 1989; 118: 85–99.
Ghetie M-A, Tucker K, Richardson J, Uhr JW, Vitetta ES. The antitumor activity of an anti-CD22 immunotoxin in SCID mice with disseminated Daudi lymphoma is enhanced by either an anti-CD19 antibody or an anti-CD19 immunotoxin. Blood 1992; 80: 2315–2320.
Colombel M, Symmans F, Gil S, O'Toole KM, Chopin D, Benson M, et al. Detection of the apoptosis-suppressing oncoprotein BCL-2 in hormone-refractory human prostate cancers. Am J Pathol 1993; 143: 390–400.
Walton MI, Whysong D, O'Connor PM, Hockenbery D, Korsmeyer SJ, Kohn KW. Constitutive expression of human BCL-2 modulates nitrogen mustard and camptothecin induced apoptosis. Cancer Res 1993; 53: 1853–1861.
Reed JC. Regulation of apoptosis by BCL-2 family proteins and its role in cancer and chemoresistance. Curr Opin Oncol 1995; 7: 541–546.
Brinkmann U, Brinkmann, E, Gallo M, Sherf U, Pastan I. Role of CAS, a human homologue to the yeast chromosome segregation gene CSE1, in toxin and TNF mediated apoptosis. Biochemistry 1996; 35: 2891–2899.
Brinkmann U, Brinkmann E, Gallo M, Pastan I. Cloning and characterzation of a cellular apoptosis susceptibility gene, the human homologue to the yeast chromosome segregation gene CSE1. Proc Natl Acad Sci USA 1995; 92: 10427–10431.
Brinkmann U. Recombinant toxins: Protein engineering for Cancer Therapy. Mol Med Today 1996; 2: 439–446.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Brinkmann, U., Mansfield, E. & Pastan, I. Effects of BCL-2 overexpression on the sensitivity of MCF-7 breast cancer cells to ricin, diphtheria and Pseudomonas toxin and immunotoxins. Apoptosis 2, 192–198 (1997). https://doi.org/10.1023/A:1026468532413
Issue Date:
DOI: https://doi.org/10.1023/A:1026468532413