Abstract
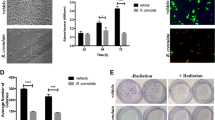
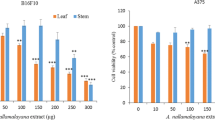
Skin cancers are the most common cancers in the world and among the different types of skin cancers, melanoma is the deadliest and incidence is rising. Previous studies have shown promising in vitro and human evidence of kiwifruit exhibiting anti-cancer effects. This study was designed to investigate if kiwifruit extract (KE) has any effect on CRL-11147 melanoma cancer cells and to investigate the possible mechanisms behind the results. The effects of KE on CRL-11147 melanoma cell survival, proliferation, and apoptosis was investigated using clonogenic survival assay, cell proliferation, and caspase-3 activity kits. Potential anti-tumor molecular mechanisms were elucidated using RT-PCR and IHC. Addition of KE decreased CRL-11147 cell colonies percentages indicated by a decreased optical density value of cancer cells when compared to control. Furthermore, treatment with KE increased relative caspase-3 activity in cancer cells, which indicated increased apoptosis of cancer cells. The anti-proliferative effect of KE on cancer cells corresponded with decreased expression of the pro-proliferative molecule Cyclin E and CDK4, while increased expression of the pro-apoptotic molecule TRAILR1 corresponded with the pro-apoptotic effect. KE decreases CRL-11147 melanoma cell growth via downregulation of Cyclin E and CDK4 and upregulation in TRAILR1. Our study suggests a potential use for KE in treatment of melanoma.






Similar content being viewed by others
References
Siegel RL, Miller KD, Jemal A. Cancer statistics, 2020. CA Cancer J Clin. 2020;70(1):7–30. https://doi.org/10.3322/caac.21590.
Rainov NG, Burkert W. Miliary brain metastases from malignant melanoma. Zentralbl Neurochir. 1996;57(1):20–4.
Barnholtz-Sloan JS, Sloan AE, Davis FG, Vigneau FD, Lai P, Sawaya RE. Incidence proportions of brain metastases in patients diagnosed (1973 to 2001) in the Metropolitan Detroit Cancer Surveillance System. J Clin Oncol. 2004;22(14):2865–72. https://doi.org/10.1200/JCO.2004.12.149.
Chiarion-Sileni V, Guida M, Ridolfi L, Romanini A, Del Bianco P, Pigozzo J, et al. Central nervous system failure in melanoma patients: results of a randomised, multicentre phase 3 study of temozolomide-and dacarbazine-based regimens. Br J Cancer. 2011;104(12):1816–21.
Sul J, Posner JB. Brain metastases: epidemiology and pathophysiology. Brain metastases. New York: Springer; 2007. p. 1–21.
Trucco LD, Mundra PA, Hogan K, Garcia-Martinez P, Viros A, Mandal AK, et al. Ultraviolet radiation-induced DNA damage is prognostic for outcome in melanoma. Nat Med. 2019;25(2):221–4.
Cohen JV, Tawbi H, Margolin KA, Amravadi R, Bosenberg M, Brastianos PK, et al. Melanoma central nervous system metastases: current approaches, challenges, and opportunities. Pigment Cell Melanoma Res. 2016;29(6):627–42. https://doi.org/10.1111/pcmr.12538.
Livingstone A, Agarwal A, Stockler MR, Menzies AM, Howard K, Morton RL. Preferences for immunotherapy in melanoma: a systematic review. Ann Surg Oncol. 2020;27(2):571–84. https://doi.org/10.1245/s10434-019-07963-y.
Rubio-Rodriguez D, De Diego BS, Perez M, Rubio-Terres C. Cost-effectiveness of drug treatments for advanced melanoma: a systematic literature review. Pharmacoeconomics. 2017;35(9):879–93. https://doi.org/10.1007/s40273-017-0517-1.
Lippi G, Mattiuzzi C. Kiwifruit and cancer: an overview of biological evidence. Nutr Cancer. 2020;72(4):547–53. https://doi.org/10.1080/01635581.2019.1650190.
Johnson GE, Ivanov VN, Hei TK. Radiosensitization of melanoma cells through combined inhibition of protein regulators of cell survival. Apoptosis. 2008;13(6):790.
Wu F, Cui L. Resveratrol suppresses melanoma by inhibiting NF-kappaB/miR-221 and inducing TFG expression. Arch Dermatol Res. 2017;309(10):823–31. https://doi.org/10.1007/s00403-017-1784-6.
Fang Y, Bradley MJ, Cook KM, Herrick EJ, Nicholl MB. A potential role for resveratrol as a radiation sensitizer for melanoma treatment. J Surg Res. 2013;183(2):645–53.
Fang Y, Chen X, Bai Q, Qin C, Mohamud AO, Zhu Z, et al. IL-9 inhibits HTB-72 melanoma cell growth through upregulation of p21 and TRAIL. J Surg Oncol. 2015;111(8):969–74.
Fang Y, DeMarco VG, Nicholl MB. Resveratrol enhances radiation sensitivity in prostate cancer by inhibiting cell proliferation and promoting cell senescence and apoptosis. Cancer Sci. 2012;103(6):1090–8. https://doi.org/10.1111/j.1349-7006.2012.02272.x.
Zhu Z, Davidson KT, Brittingham A, Wakefield MR, Bai Q, Xiao H, et al. Trichomonas vaginalis: a possible foe to prostate cancer. Med Oncol. 2016;33(10):115. https://doi.org/10.1007/s12032-016-0832-y.
Johnson DG, Walker CL. Cyclins and cell cycle checkpoints. Annu Rev Pharmacol Toxicol. 1999;39:295–312. https://doi.org/10.1146/annurev.pharmtox.39.1.295.
Sherr CJ, Roberts JM. Living with or without cyclins and cyclin-dependent kinases. Genes Dev. 2004;18(22):2699–711.
Zhang D, Li X, Chen C, Li Y, Zhao L, Jing Y, et al. Attenuation of p38-mediated miR-1/133 expression facilitates myoblast proliferation during the early stage of muscle regeneration. PLoS ONE. 2012;7(7):e41478. https://doi.org/10.1371/journal.pone.0041478.
Zhu Z, Zhang D, Lee H, Menon AA, Wu J, Hu K, et al. Macrophage-derived apoptotic bodies promote the proliferation of the recipient cells via shuttling microRNA-221/222. J Leukoc Biol. 2017;101(6):1349–59. https://doi.org/10.1189/jlb.3A1116-483R.
Donnellan R, Chetty R. Cyclin E in human cancers. FASEB J. 1999;13(8):773–80. https://doi.org/10.1096/fasebj.13.8.773.
Kollmann K, Briand C, Bellutti F, Schicher N, Blunder S, Zojer M, et al. The interplay of CDK4 and CDK6 in melanoma. Oncotarget. 2019;10(14):1346–59. https://doi.org/10.18632/oncotarget.26515.
Zhao H, Zheng B. Dual targeting of autophagy and MEK in KRAS mutant cancer. Trends Cancer. 2019;5(6):327–9. https://doi.org/10.1016/j.trecan.2019.04.003.
Bartkova J, Lukas J, Guldberg P, Alsner J, Kirkin AF, Zeuthen J, et al. The p16-cyclin D/Cdk4-pRb pathway as a functional unit frequently altered in melanoma pathogenesis. Can Res. 1996;56(23):5475–83.
Hussussian CJ, Struewing JP, Goldstein AM, Higgins PA, Ally DS, Sheahan MD, et al. Germline p16 mutations in familial melanoma. Nat Genet. 1994;8(1):15–21. https://doi.org/10.1038/ng0994-15.
Sharpless NE, Chin L. The INK4a/ARF locus and melanoma. Oncogene. 2003;22(20):3092–8.
Wajant H. TRAIL-and TNF-induced signaling complexes—so similar yet so different. EMBO J. 2017;36(9):1117–9.
Gerspach J, Pfizenmaier K, Wajant H. Therapeutic targeting of CD95 and the TRAIL death receptors. Recent Pat Anticancer Drug Discov. 2011;6(3):294–310. https://doi.org/10.2174/157489211796957739.
Oka K, Suzuki Y, Nakano T. Expression of p27 and p53 in cervical squamous cell carcinoma patients treated with radiotherapy alone: radiotherapeutic effect and prognosis. Cancer. 2000;88(12):2766–73. https://doi.org/10.1002/1097-0142(20000615)88:12%3c2766::aid-cncr15%3e3.0.co;2-g.
McKean MA, Amaria RN. Multidisciplinary treatment strategies in high-risk resectable melanoma: role of adjuvant and neoadjuvant therapy. Cancer Treat Rev. 2018;70:144–53. https://doi.org/10.1016/j.ctrv.2018.08.011.
Kim MM, Parmar H, Cao Y, Pramanik P, Schipper M, Hayman J, et al. Whole brain radiotherapy and RRx-001: two partial responses in radioresistant melanoma brain metastases from a phase I/II clinical trial: a TITE-CRM phase I/II clinical trial. Transl Oncol. 2016;9(2):108–13. https://doi.org/10.1016/j.tranon.2015.12.003.
Singh M, Alavi A, Wong R, Akita S. Radiodermatitis: a review of our current understanding. Am J Clin Dermatol. 2016;17(3):277–92.
Acknowledgements
This work was supported by grants from Des Moines University for Yujiang Fang.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no disclosures.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kou, L., Zhu, Z., Redington, C. et al. Potential use of kiwifruit extract for treatment of melanoma. Med Oncol 38, 25 (2021). https://doi.org/10.1007/s12032-021-01465-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12032-021-01465-2