Abstract
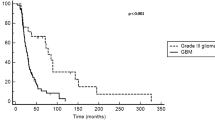
Our objective is to assess treatment efficacy, safety and pattern of response and recurrence in patients with recurrent high-grade glioma treated with bevacizumab and irinotecan. We reviewed retrospectively 51 patients with recurrent high-grade glioma treated with this combination at the Henry Ford Hermelin Brain Tumor Center from 11/15/2005 to 04/01/2008. The 6-month progression-free survival (PFS) for anaplastic gliomas (AGs) was 78.6 and 63.7% for glioblastoma. The median PFS was 13.4 months for AG and 7.6 months for those with glioblastoma. The overall survival rate (OS) at 6 months was 85.7% for AG and 78.0% for glioblastoma. The 12-month OS was 77.9% for AG and 42.6% for glioblastoma. The median OS time for AGs was not reached and was 11.5 months for those with glioblastoma. Thirty-six out of 51 (70.59%) patients demonstrated partial (32/51) or complete (4/51) radiographic response to treatment and 8/51 (15.69%) remained stable. Of the 38 who demonstrated progression on post-gadolinium studies, 23 showed distant progression with or without local recurrence. Seven patients showed progression on FLAIR without concordant findings on post-Gd sequences. Six patients (11.76%) discontinued treatment due to a treatment-emergent adverse event, including one with end-stage renal failure and another with gastric perforation. No symptomatic intracranial hemorrhages were reported. Patients with recurrent high-grade glioma treated with bevacizumab plus irinotecan demonstrate an excellent radiographic response rate and improved clinical outcome when compared to historical data. The high rate of distant tumor progression suggests that tumors may adapt to inhibition of angiogenesis by increased infiltration and vascular co-option.





Similar content being viewed by others
References
Stupp R, Mason WP, van den Bent MJ et al (2005) Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N Engl J Med 352:987–996
Wong ET, Hess KR, Gleason MJ et al (1999) Outcomes and prognostic factors in recurrent glioma patients enrolled onto phase II clinical trials. J Clin Oncol 17:2572–2578
Lacroix M, Abi-Said D, Fourney DR et al (2001) A multivariate analysis of 416 patients with glioblastoma multiforme: prognosis, extent of resection, and survival. J Neurosurg 95:190–198
Friedman HS, Petros WP, Friedman AH, Schaaf LJ, Kerby T, Lawyer J et al (1999) Irinotecan therapy in adults with recurrent or progressive malignant glioma. J Clin Oncol 17:1516–1525
Kargiotis O, Rao JS, Kyritsis AP (2006) Mechanism of angiogenesis in gliomas. J Neurooncol 78:281–283
Rubenstein JL, Kim J, Ozaka T (2000) Anti-VEGF antibody treatment of glioblastoma prolongs survival but results in increased vascular cooption. Neoplasia 2:306–314
Ferrara N, Gerber HP, LeCouter J (2003) The biology of VEGF and its receptors. Nat Med 9:669–676
Hurwitz H, Fehrenbacker L, Novotny W et al (2004) Bevacizumab plus irinotecan, fluorouracil and leucovorin for metastatic colorectal cancer. N Engl J Med 350:2335–2342
Vredenburgh JJ, Desjardins A, Herndon JE et al (2007) Phase II trial of bevacizumab and irinotecan in recurrent malignant glioma. Clin Cancer Res 13:1253–1259
Norden AD, Young GS, Setayesh K et al (2008) Bevacizumab for recurrent malignant gliomas: efficacy, toxicity, and patterns of recurrence. Neurology 70:779–787
Wong ET, Brem S (2007) Taming glioblastoma: targeting angiogenesis. J Clin Oncol 26:4705–4706
Claes A, Gambarota G, Hamans B et al (2008) Magnetic resonance imaging-based detection of glial brain tumors in mice after antiangiogenic treatment. Int J Cancer 22:1981–1986
Cloughesy T, Prados M, Wen P, Mikkelsen T et al (2008) Randomized phase II study of bevacizumab alone or in combination with irinotecan. J Clin Oncol, ASCO Annual meeting proceedings (Post-meeting edn), vol 26, no 15S (May 20 Suppl), 2010b
MacDonald DR, Cascino TL, Schold SC Jr et al (1990) Response criteria for phase II studies of supratentorial malignant glioma. J Clin Oncol 8:1277–1280
Pope WB, Lai A, Nghimephu P (2006) MRI in patients with high-grade gliomas treated with bevacizumab and chemotherapy. Neurology 66:1258–1260
Fiveash JB, Spencer SA (2005) Role of radiation therapy and radiosurgery in glioblastoma multiforme. In: Markert J, DeVita V, Rosenberg S (eds) Glioblastoma multiforme. Jones and Bartlett Publishers, Sudbury, pp 146–147
Bao S, Wu Q, Sathornsumetee S et al (2006) Stem cell-like glioma cells promote tumor angiogenesis through vascular endothelial growth factor. Cancer Res 66:7843–7848
Essig M, Metzner R, Bonsanto M (2001) Postoperative fluid-attenuated inversion recovery MR imaging of cerebral gliomas: initial results. Eur Radiol 11:2004–2010
Jansen M, de Witt Hamer PC, Witmer AN, Troost D, van Noorden CJ (2004) Current perspectives on antiangiogenesis strategies in the treatment of malignant gliomas. Brain Res Brain Res Rev 45:146–163
Yung WK, Albright RE, Olson J et al (2000) A phase II study of temozolomide vs. procarbazine in patients with glioblastoma multiforme at first relapse. Br J Cancer 83:588–593
McConville P, Hambardzumyan D, Moody JB et al (2007) Magnetic resonance imaging determination of tumor grade and early response to temozolomide in a genetically engineered mouse model of glioma. Clin Cancer Res 13:2897–2904
Baehring JM, Linda W, Bannykj S et al (2007) Diffusion MRI in the early diagnosis of malignant glioma. J Neurooncol 82:221–225
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zuniga, R.M., Torcuator, R., Jain, R. et al. Efficacy, safety and patterns of response and recurrence in patients with recurrent high-grade gliomas treated with bevacizumab plus irinotecan. J Neurooncol 91, 329–336 (2009). https://doi.org/10.1007/s11060-008-9718-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-008-9718-y