Abstract
Background and Aims
Pigment epithelium-derived factor (PEDF) has been shown to be a potent inhibitor of inflammation through its anti-oxidative property. Since oxidative response is considered to play the pivotal role of the development and progression of nonalcoholic steatohepatitis (NASH), it is conceivable that PEDF may play a protective role against NASH. In this study, we examined whether administration of PEDF slowed the progression of NASH in mice models.
Methods
Mice were fed methionine- and choline-deficient (MCD) diet with or without intramuscular administration of adenovirus-expressing PEDF (Ad-PEDF). Effects of PEDF administration on NASH were histologically and biochemically evaluated.
Results
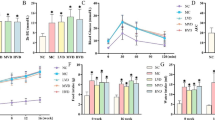
Administration of Ad-PEDF significantly decreased hepatic fat storage as well as serum levels of ALT in MCD diet-fed mice. Dihydroethidium staining showed that MCD diet-triggered oxidative stress was reduced in the liver of Ad-PEDF-administered mice compared to that of PBS- or Ad-LacZ-administered mice. Activation of Kupffer cells and hepatic fibrosis was also inhibited by Ad-PEDF administration. Quantitative real-time RT-PCR revealed that MCD diet up-regulated expressions of TNF-α, IL-1β, IL-6, TGF-β, collagen-1, and collagen-3 mRNA, which were also attenuated with Ad-PEDF administration, whereas MCD diet-induced down-regulation of expressions of PPAR-γ mRNA was restored with Ad-PEDF administration. Furthermore, immunoblotting analysis showed that MCD diet-induced up-regulation of NADPH oxidase components was significantly decreased in Ad-PEDF-administered mice.
Conclusions
The present results demonstrated for the first time that PEDF could slow the development and progression of steatohepatitis through the suppression of steatosis and inflammatory response in MCD diet-fed mice. Our study suggests that PEDF supplementation may be a novel therapeutic strategy for the treatment of NASH.





Similar content being viewed by others
References
Green RM. NASH—hepatic metabolism and not simply the metabolic syndrome. Hepatology. 2003;38:14–17.
Fabbrini E, Sullivan S, Klein S. Obesity and nonalcoholic fatty liver disease: biochemical, metabolic, and clinical implications. Hepatology. 2010;51:679–689.
Tilg H, Moschen AR. Evolution of inflammation in nonalcoholic fatty liver disease: the multiple parallel hits hypothesis. Hepatology. 2010;52:1836–1846.
Tombran-Tink J, Chader GG, Johnson LV. PEDF: a pigment epithelium-derived factor with potent neuronal differentiative activity. Exp Eye Res. 1991;53:411–414.
Dawson DW, Volpert OV, Gillis P, et al. Pigment epithelium-derived factor: a potent inhibitor of angiogenesis. Science. 1999;285:245–248.
Yamagishi S, Adachi H, Abe A, et al. Elevated serum levels of pigment epithelium-derived factor in the metabolic syndrome. J Clin Endocrinol Metab. 2006;91:2447–2450.
Tahara N, Yamagishi S, Tahara A, et al. Serum level of pigment epithelium-derived factor is a marker of atherosclerosis in humans. Atherosclerosis. 2011;219:311–315.
Yoshida T, Yamagishi S, Nakamura K, et al. Pigment epithelium-derived factor (PEDF) ameliorates advanced glycation end product (AGE)-induced hepatic insulin resistance in vitro by suppressing Rac-1 activation. Horm Metab Res. 2008;40:620–625.
Matsui T, Nishino Y, Ojima A, Maeda S, Tahara N, Yamagishi S. Pigment epithelium-derived factor improves metabolic derangements and ameliorates dysregulation of adipocytokines in obese type 2 diabetic rats. Am J Pathol. 2014;184:1094–1103.
Notari L, Baladron V, Aroca-Aguilar JD, et al. Identification of a lipase-linked cell membrane receptor for pigment epithelium-derived factor. J Biol Chem. 2006;281:38022–38037.
Chung C, Doll JA, Gattu AK, et al. Anti-angiogenic pigment epithelium-derived factor regulates hepatocyte triglyceride content through adipose triglyceride lipase (ATGL). J Hepatol. 2008;48:471–478.
Wu JW, Wang SP, Alvarez F, et al. Deficiency of liver adipose triglyceride lipase in mice causes progressive hepatic steatosis. Hepatology. 2011;54:122–132.
Nakamura K, Yamagishi S, Matsui T, et al. Pigment epithelium-derived factor inhibits neointimal hyperplasia after vascular injury by blocking NADPH oxidase-mediated reactive oxygen species generation. Am J Pathol. 2007;170:2159–2170.
Bligh EG, Dyer WJ. A rapid method of total lipid extraction and purification. Can J Biochem Physiol. 1959;37:911–917.
Hashimoto M, Tanabe Y, Fujii Y, Kikuta T, Shibata H, Shido O. Chronic administration of docosahexaenoic acid ameliorates the impairment of spatial cognition learning ability in amyloid beta-infused rats. J Nutr. 2005;135:549–555.
Smirnova E, Goldberg EB, Makarova KS, Lin L, Brown WJ, Jackson CL. ATGL has a key role in lipid droplet/adiposome degradation in mammalian cells. EMBO Rep. 2006;7:106–113.
Paik YH, Brenner DA. NADPH oxidase mediated oxidative stress in hepatic fibrogenesis. Korean J Hepatol. 2011;17:251–257.
De Minicis S, Seki E, Paik YH, et al. Role and cellular source of nicotinamide adenine dinucleotide phosphate oxidase in hepatic fibrosis. Hepatology. 2010;52:1420–1430.
Bettaieb A, Jiang JX, Sasaki Y, et al. Hepatocyte nicotinamide adenine dinucleotide phosphate reduced oxidase 4 regulates stress signaling, fibrosis, and insulin sensitivity during development of steatohepatitis in mice. Gastroenterology. 2015;149:468–480.
Yoshida T, Yamagishi S, Nakamura K, et al. Pigment epithelium-derived factor (PEDF) blocks the interleukin-6 signaling to C-reactive protein expression in Hep3B cells by suppressing Rac-1 activation. Life Sci. 2006;79:1981–1987.
Mejias M, Coch L, Berzigotti A, et al. Antiangiogenic and antifibrogenic activity of pigment epithelium-derived factor (PEDF) in bile duct-ligated portal hypertensive rats. Gut. 2015;64:657–666.
Cui W, Matsuno K, Iwata K, et al. NOX1/nicotinamide adenine dinucleotide phosphate, reduced form (NADPH) oxidase promotes proliferation of stellate cells and aggravates liver fibrosis induced by bile duct ligation. Hepatology. 2011;54:949–958.
Tanaka N, Takahashi S, Zhang Y, et al. Role of fibroblast growth factor 21 in the early stage of NASH induced by methionine- and choline-deficient diet. Biochem Biophys Acta. 2015;1852:1242–1252.
Takahashi Y, Soejima Y, Kumagai A, Watanabe M, Uozaki H, Fukusato T. Inhibitory effects of Japanese herbal medicines sho-saiko-to and juzen-taiho-to on nonalcoholic steatohepatitis in mice. PLoS ONE. 2014;9:e87279.
Tomita K, Teratani T, Suzuki T, et al. Free cholesterol accumulation in hepatic stellate cells: mechanism of liver fibrosis aggravation in nonalcoholic steatohepatitis in mice. Hepatology. 2014;59:154–169.
Wu CW, Chu ES, Lam CN, et al. PPARγ is essential for protection against nonalcoholic steatohepatitis. Gene Ther. 2010;17:790–798.
Yu J, Zhang S, Chu ES, et al. Peroxisome proliferator-activated receptors gamma reverses hepatic nutritional fibrosis in mice and suppresses activation of hepatic stellate cells in vitro. Int J Biochem Cell Biol. 2010;42:948–957.
Hirsch J, Johnson CL, Nelius T, Kennedy R, Riese W, Filleur S. PEDF inhibits IL8 production in prostate cancer cells through PEDF receptor/phospholipase A2 and regulation of NFκB and PPARγ. Cytokine. 2011;55:202–210.
Blanquicett C, Kang BY, Ritzenthaler JD, Jones DP, Hart CM. Oxidative stress modulates PPARγ in vascular endothelial cells. Free Radic Biol Med. 2010;48:1618–1625.
Ho TC, Chen SL, Yang YC, Liao CL, Cheng HC, Tsao YP. PEDF induces p53-mediated apoptosis through PPAR gamma signaling in human umbilical vein endothelial cells. Cardiovasc Res. 2007;76:213–223.
Ishibashi Y, Matsui T, Ohta K, et al. PEDF inhibits AGE-induced podocyte apoptosis via PPAR-gamma activation. Microvasc Res. 2013;85:54–58.
Nakamura K, Yamagishi S, Yoshida T, et al. Hydrogen peroxide stimulates pigment epithelium-derived factor gene and protein expression in the human hepatocyte cell line OUMS-29. J Int Med Res. 2007;35:427–432.
Hyogo H, Yamagishi S, Maeda S, Kimura Y, Ishitobi T, Chayama K. Serum levels of pigment epithelium-derived factor (PEDF) are independently associated with procollagen III N-terminal peptide levels in patients with nonalcoholic fatty liver disease. Clin Biochem. 2012;45:1554–1557.
Acknowledgments
We thank Yasuko Imamura and Masako Hayakawa for excellent technical help and Taeko Narisawa for secretarial assistance. This work was supported by JSPS Grant-in-Aid for Scientific Research (C) Grant Number 16K09385 (TT).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no financial conflicts of interest.
Rights and permissions
About this article
Cite this article
Yoshida, T., Akiba, J., Matsui, T. et al. Pigment Epithelium-Derived Factor (PEDF) Prevents Hepatic Fat Storage, Inflammation, and Fibrosis in Dietary Steatohepatitis of Mice. Dig Dis Sci 62, 1527–1536 (2017). https://doi.org/10.1007/s10620-017-4550-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-017-4550-x