Abstract
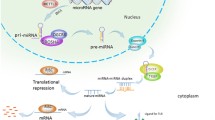
MicroRNAs (miRNAs) are small noncoding, double-stranded RNA molecules that can mediate the expression of target genes with complementary sequences. About 5,300 human genes have been implicated as targets for miRNAs, making them one of the most abundant classes of regulatory genes in humans. MiRNAs recognize their target mRNAs based on sequence complementarity and act on them to cause the inhibition of protein translation by degradation of mRNA. Besides contributing to development and normal function, microRNAs have functions in various human diseases. Given the importance of miRNAs in regulating cellular differentiation and proliferation, it is not surprising that their misregulation is linked to cancer. In cancer, miRNAs function as regulatory molecules, acting as oncogenes or tumor suppressors. Amplification or overexpression of miRNAs can down-regulate tumor suppressors or other genes involved in cell differentiation, thereby contributing to tumor formation by stimulating proliferation, angiogenesis, and invasion; i.e., they act as oncogenes. Similarly, miRNAs can down-regulate different proteins with oncogenic activity; i.e., they act as tumor suppressors. This review will highlight the recent discoveries regarding miRNAs and their importance in cancer.


Similar content being viewed by others
References
Lewis, B. P., Burge, C. B., & Bartel, D. P. (2005). Conserved seed pairing, often flanked by adenosines, indicates that thousands of human genes are microRNA targets. Cell, 120(1), 15–20.
Rajewsky, N. (2006). microRNA target predictions in animals. Nature Genetics, 38(Suppl), S8–S13.
Lee, R. C., Feinbaum, R. L., & Ambros, V. (1993). The C. elegans heterochronic gene lin-4 encodes small RNAs with antisense complementarity to lin-14. Cell, 75(5), 843–854.
Reinhart, B. J., et al. (2000). The 21-nucleotide let-7 RNA regulates developmental timing in Caenorhabditis elegans. Nature, 403(6772), 901–906.
Pasquinelli, A. E., et al. (2000). Conservation of the sequence and temporal expression of let-7 heterochronic regulatory RNA. Nature, 408(6808), 86–89.
Lagos-Quintana, M., et al. (2001). Identification of novel genes coding for small expressed RNAs. Science, 294(5543), 853–858.
Lau, N. C., et al. (2001). An abundant class of tiny RNAs with probable regulatory roles in Caenorhabditis elegans. Science, 294(5543), 858–862.
Lee, R. C., & Ambros, V. (2001). An extensive class of small RNAs in Caenorhabditis elegans. Science, 294(5543), 862–864.
ALTMAN, L.K., 5 Pioneering Scientists Win Lasker Medical Prizes in New York Times. 2008
Bagga, S., et al. (2005). Regulation by let-7 and lin-4 miRNAs results in target mRNA degradation. Cell, 122(4), 553–563.
Pillai, R. S. (2005). MicroRNA function: multiple mechanisms for a tiny RNA? RNA, 11(12), 1753–1761.
Bernstein, E., et al. (2003). Dicer is essential for mouse development. Nature Genetics, 35(3), 215–217.
Harris, K. S., et al. (2006). Dicer function is essential for lung epithelium morphogenesis. Proceedings of the National Academy of Sciences of the United States of America, 103(7), 2208–2213.
Harfe, B. D., et al. (2005). The RNaseIII enzyme Dicer is required for morphogenesis but not patterning of the vertebrate limb. Proceedings of the National Academy of Sciences of the United States of America, 102(31), 10898–10903.
O'Rourke, J. R., et al. (2007). Essential role for Dicer during skeletal muscle development. Developmental Biology, 311(2), 359–368.
Muljo, S. A., et al. (2005). Aberrant T cell differentiation in the absence of Dicer. Journal of Experimental Medicine, 202(2), 261–269.
Yi, R., et al. (2008). A skin microRNA promotes differentiation by repressing ‘stemness’. Nature, 452(7184), 225–229.
Ashraf, S. I., & Kunes, S. (2006). A trace of silence: memory and microRNA at the synapse. Current Opinion in Neurobiology, 16(5), 535–539.
Poy, M. N., et al. (2004). A pancreatic islet-specific microRNA regulates insulin secretion. Nature, 432(7014), 226–230.
Esau, C., et al. (2006). miR-122 regulation of lipid metabolism revealed by in vivo antisense targeting. Cell Metabolic, 3(2), 87–98.
Esau, C. C., & Monia, B. P. (2007). Therapeutic potential for microRNAs. Advanced drug delivery reviews, 59(2–3), 101–114.
Care, A., et al. (2007). MicroRNA-133 controls cardiac hypertrophy. Nature Medicine, 13(5), 613–618.
Xiao, C., & Rajewsky, K. (2009). MicroRNA control in the immune system: basic principles. Cell, 136(1), 26–36.
Cameron, J. E., et al. (2008). Epstein-Barr virus latent membrane protein 1 induces cellular MicroRNA miR-146a, a modulator of lymphocyte signaling pathways. Journal of Virology, 82(4), 1946–1958.
Gottwein, E., et al. (2007). A viral microRNA functions as an orthologue of cellular miR-155. Nature, 450(7172), 1096–1099.
Moschos, S. A., et al. (2007). Expression profiling in vivo demonstrates rapid changes in lung microRNA levels following lipopolysaccharide-induced inflammation but not in the anti-inflammatory action of glucocorticoids. BMC Genomics, 8, 240.
Sonkoly, E., Stahle, M., & Pivarcsi, A. (2008). MicroRNAs and immunity: novel players in the regulation of normal immune function and inflammation. Seminars in Cancer Biology, 18(2), 131–140.
Calin, G. A., et al. (2002). Frequent deletions and down-regulation of micro-RNA genes miR15 and miR16 at 13q14 in chronic lymphocytic leukemia. Proceedings of the National Academy of Sciences of the United States of America, 99(24), 15524–15529.
Jiang, J., et al. (2005). Real-time expression profiling of microRNA precursors in human cancer cell lines. Nucleic Acids Research, 33(17), 5394–5403.
Davison, T. S., Johnson, C. D., & Andruss, B. F. (2006). Analyzing micro-RNA expression using microarrays. Methods in Enzymology, 411, 14–34.
Liang, Z., et al. (2007). Blockade of invasion and metastasis of breast cancer cells via targeting CXCR4 with an artificial microRNA. Biochemical and Biophysical Research Communications, 363(3), 542–546.
Miska, E. A. (2005). How microRNAs control cell division, differentiation and death. Current Opinion in Genetics and Development, 15(5), 563–568.
Lee, Y. S., et al. (2005). Depletion of human micro-RNA miR-125b reveals that it is critical for the proliferation of differentiated cells but not for the down-regulation of putative targets during differentiation. Journal of Biological Chemistry, 280(17), 16635–16641.
Roldo, C., et al. (2006). MicroRNA expression abnormalities in pancreatic endocrine and acinar tumors are associated with distinctive pathologic features and clinical behavior. Journal of Clinical Oncology, 24(29), 4677–4684.
Dillhoff, M., et al. (2008). MicroRNA-21 is overexpressed in pancreatic cancer and a potential predictor of survival. Journal of Gastrointestinal Surgery, 12(12), 2171–2176.
Gironella, M., et al. (2007). Tumor protein 53-induced nuclear protein 1 expression is repressed by miR-155, and its restoration inhibits pancreatic tumor development. Proceedings of the National Academy of Sciences of the United States of America, 104(41), 16170–16175.
Burk, U., et al. (2008). A reciprocal repression between ZEB1 and members of the miR-200 family promotes EMT and invasion in cancer cells. EMBO Reports, 9(6), 582–589.
Nakajima, G., et al. (2006). Non-coding microRNAs hsa-let-7g and hsa-miR-181b are associated with Chemoresponse to S-1 in Colon Cancer. Cancer Genomics Proteomics, 3(5), 317–324.
Lanza, G., et al. (2007). mRNA/microRNA gene expression profile in microsatellite unstable colorectal cancer. Molecular Cancer, 6, 54.
Asangani, I. A., et al. (2008). MicroRNA-21 (miR-21) post-transcriptionally downregulates tumor suppressor Pdcd4 and stimulates invasion, intravasation and metastasis in colorectal cancer. Oncogene, 27(15), 2128–2136.
Hayashita, Y., et al. (2005). A polycistronic microRNA cluster, miR-17–92, is overexpressed in human lung cancers and enhances cell proliferation. Cancer Research, 65(21), 9628–9632.
Matsubara, H., et al. (2007). Apoptosis induction by antisense oligonucleotides against miR-17–5p and miR-20a in lung cancers overexpressing miR-17–92. Oncogene, 26(41), 6099–6105.
Chang, T. C., et al. (2007). Transactivation of miR-34a by p53 broadly influences gene expression and promotes apoptosis. Molecular Cell, 26(5), 745–752.
Lodygin, D., et al. (2008). Inactivation of miR-34a by aberrant CpG methylation in multiple types of cancer. Cell Cycle, 7(16), 2591–2600.
Michael, M. Z., et al. (2003). Reduced accumulation of specific microRNAs in colorectal neoplasia. Molecular Cancer Research, 1(12), 882–891.
Akao, Y., Nakagawa, Y., & Naoe, T. (2006). MicroRNAs 143 and 145 are possible common onco-microRNAs in human cancers. Oncology Reports, 16(4), 845–850.
Shi, B., et al. (2007). Micro RNA 145 targets the insulin receptor substrate-1 and inhibits the growth of colon cancer cells. Journal of Biological Chemistry, 282(45), 32582–32590.
Akao, Y., Nakagawa, Y., & Naoe, T. (2006). Let-7 microRNA functions as a potential growth suppressor in human colon cancer cells. Biological and Pharmaceutical Bulletin, 29(5), 903–906.
Takamizawa, J., et al. (2004). Reduced expression of the let-7 microRNAs in human lung cancers in association with shortened postoperative survival. Cancer Research, 64(11), 3753–3756.
Johnson, S. M., et al. (2005). RAS is regulated by the let-7 microRNA family. Cell, 120(5), 635–647.
Johnson, C. D., et al. (2007). The let-7 microRNA represses cell proliferation pathways in human cells. Cancer Research, 67(16), 7713–7722.
Lee, Y. S., & Dutta, A. (2007). The tumor suppressor microRNA let-7 represses the HMGA2 oncogene. Genes and Development, 21(9), 1025–1030.
Bommer, G. T., et al. (2007). p53-mediated activation of miRNA34 candidate tumor-suppressor genes. Current Biology, 17(15), 1298–1307.
Calin, G. A., & Croce, C. M. (2006). MicroRNA-cancer connection: the beginning of a new tale. Cancer Research, 66(15), 7390–7394.
Cimmino, A., et al. (2005). miR-15 and miR-16 induce apoptosis by targeting BCL2. Proceedings of the National Academy of Sciences of the United States of America, 102(39), 13944–13949.
Lin, T., et al. (2009). MicroRNA-143 as a tumor suppressor for bladder cancer. The Journal of Urology, 181, 1372–1380.
Varambally, S., et al. (2008). Genomic loss of microRNA-101 leads to overexpression of histone methyltransferase EZH2 in cancer. Science, 322(5908), 1695–1699.
Liu, C. G., et al. (2004). An oligonucleotide microchip for genome-wide microRNA profiling in human and mouse tissues. Proceedings of the National Academy of Sciences of the United States of America, 101(26), 9740–9744.
Walsh, T., & King, M. C. (2007). Ten genes for inherited breast cancer. Cancer Cell, 11(2), 103–105.
Xin, F., et al. (2008). Computational analysis of MicroRNA profiles and their target genes suggests significant involvement in breast cancer antiestrogen resistance. Bioinformatics, 25, 430–434.
Scott, G. K., et al. (2006). Rapid alteration of microRNA levels by histone deacetylase inhibition. Cancer Research, 66(3), 1277–1281.
Abdelrahim, M., et al. (2002). Small inhibitory RNA duplexes for Sp1 mRNA block basal and estrogen-induced gene expression and cell cycle progression in MCF-7 breast cancer cells. Journal of Biological Chemistry, 277(32), 28815–28822.
Ma, L., Teruya-Feldstein, J., & Weinberg, R. A. (2007). Tumour invasion and metastasis initiated by microRNA-10b in breast cancer. Nature, 449(7163), 682–688.
Si, M. L., et al. (2007). miR-21-mediated tumor growth. Oncogene, 26(19), 2799–2803.
Zhu, S., et al. (2007). MicroRNA-21 targets the tumor suppressor gene tropomyosin 1 (TPM1). Journal of Biological Chemistry, 282(19), 14328–14336.
Boyd, J., et al. (1995). Regulation of microfilament organization and anchorage-independent growth by tropomyosin 1. Proceedings of the National Academy of Sciences of the United States of America, 92(25), 11534–11538.
Frankel, L. B., et al. (2008). Programmed cell death 4 (PDCD4) is an important functional target of the microRNA miR-21 in breast cancer cells. Journal of Biological Chemistry, 283(2), 1026–1033.
Hossain, A., Kuo, M. T., & Saunders, G. F. (2006). Mir-17–5p regulates breast cancer cell proliferation by inhibiting translation of AIB1 mRNA. Molecular and Cellular Biology, 26(21), 8191–8201.
Murray, G. I., et al. (1997). Tumor-specific expression of cytochrome P450 CYP1B1. Cancer Research, 57(14), 3026–3031.
Tsuchiya, Y., et al. (2006). MicroRNA regulates the expression of human cytochrome P450 1B1. Cancer Research, 66(18), 9090–9098.
Mattie, M. D., et al. (2006). Optimized high-throughput microRNA expression profiling provides novel biomarker assessment of clinical prostate and breast cancer biopsies. Molecular Cancer, 5, 24.
Scott, G. K., et al. (2007). Coordinate suppression of ERBB2 and ERBB3 by enforced expression of micro-RNA miR-125a or miR-125b. Journal of Biological Chemistry, 282(2), 1479–1486.
Eger, A., et al. (2005). DeltaEF1 is a transcriptional repressor of E-cadherin and regulates epithelial plasticity in breast cancer cells. Oncogene, 24(14), 2375–2385.
Hurteau, G. J., et al. (2007). Overexpression of the microRNA hsa-miR-200c leads to reduced expression of transcription factor 8 and increased expression of E-cadherin. Cancer Research, 67(17), 7972–7976.
Adams, B. D., Furneaux, H., & White, B. A. (2007). The micro-ribonucleic acid (miRNA) miR-206 targets the human estrogen receptor-alpha (ERalpha) and represses ERalpha messenger RNA and protein expression in breast cancer cell lines. Molecular Endocrinology, 21(5), 1132–1147.
Kondo, N., et al. (2008). miR-206 Expression is down-regulated in estrogen receptor alpha-positive human breast cancer. Cancer Research, 68(13), 5004–5008.
Chambers, A. F., Groom, A. C., & MacDonald, I. C. (2002). Dissemination and growth of cancer cells in metastatic sites. Nature Reviews Cancer, 2(8), 563–572.
Fidler, I. J. (2003). The pathogenesis of cancer metastasis: the ‘seed and soil’ hypothesis revisited. Nature Review Cancer, 3(6), 453–458.
Huang, Q., et al. (2008). The microRNAs miR-373 and miR-520c promote tumour invasion and metastasis. Nature Cell Biology, 10(2), 202–210.
Tavazoie, S. F., et al. (2008). Endogenous human microRNAs that suppress breast cancer metastasis. Nature, 451(7175), 147–152.
Yu, F., et al. (2007). Let-7 regulates self renewal and tumorigenicity of breast cancer cells. Cell, 131(6), 1109–1123.
Gebeshuber, C. A., Zatloukal, K., & Martinez, J. (2009). miR-29a suppresses tristetraprolin, which is a regulator of epithelial polarity and metastasis. EMBO Reports, 10(4), 400–405.
Acknowledgments
We apologize to all researchers whose work could not be cited in this review as a result of space constraints. This work was supported by NIH RO1 CA 115706, Susan Komen BCTR 0600278, and funds from the Louisiana Cancer Research Consortium to SKA.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Shenouda, S.K., Alahari, S.K. MicroRNA function in cancer: oncogene or a tumor suppressor?. Cancer Metastasis Rev 28, 369–378 (2009). https://doi.org/10.1007/s10555-009-9188-5
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10555-009-9188-5