Abstract
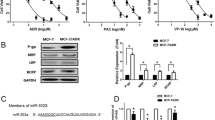
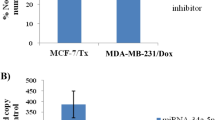
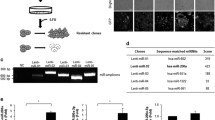
Multidrug resistance (MDR) remains one of the most significant obstacles in breast cancer treatment, and this process often involves dysregulation of a great number of microRNAs (miRNAs). Some miRNAs are indicators of drug resistance and confer resistance to chemotherapeutic drugs, although our understanding of this complex process is still incomplete. We have used a combination of miRNA profiling and real-time PCR in two drug-resistant derivatives of MCF-7 and Cal51 cells. Experimental modulation of miR expression has been obtained by retroviral transfection. Taxol and doxorubicin IC50 values were obtained by short-term drug sensitivity assays. Apoptosis was determined by flow cytometry after annexin V staining, by caspase 3/7 and caspase 9 activity assays and the levels of apoptosis-related proteins bcl-2 and bax by real-time PCR and Western blot. miR target was studied using transient transfection of luciferase constructs with the 3′ untranslated regions (UTR) of target mRNAs. Small interfering RNA-mediated genetic knock-down was performed in MDR cells and its modulatory effect on apoptosis examined. The effect of miRNA on tumorigenicity and tumor drug response was studied in mouse xenografts. miRNA profiling of two drug-resistant breast cancer cell models indicated that miR-218 was down-regulated in both MCF-7/A02 and CALDOX cells. Ectopic expression of miR-218 resensitized both drug-resistant cell lines to doxorubicin and taxol due to an increase in apoptosis. miR-218 binds survivin (BIRC5) mRNA 3′-UTR and down-regulated reporter luciferase activity. Experimental down-regulation of survivin by RNA interference in drug-resistant cells did mimic the sensitization observed when miRNA-218 was up-regulated. In addition, resensitization to taxol was also observed in mouse tumor xenografts from cells over-expressing miR-218. miR-218 is involved in the development of MDR in breast cancer cells via targeting survivin and leading to evasion of apoptosis. Targeting miR-218 and survivin may thus provide a potential strategy for reversing drug resistance in breast cancer.






Similar content being viewed by others
References
Siegel R, Naishadham D, Jemal A (2012) Cancer statistics, 2012. CA Cancer J Clin 62(1):10–29. doi:10.3322/caac.20138
Gu J, Fang X, Hao J, Sha X (2015) Reversal of P-glycoprotein-mediated multidrug resistance by CD44 antibody-targeted nanocomplexes for short hairpin RNA-encoding plasmid DNA delivery. Biomaterials 45:99–114. doi:10.1016/j.biomaterials.2014.12.030
Naci D, El Azreq MA, Chetoui N, Lauden L, Sigaux F, Charron D, Al-Daccak R, Aoudjit F (2012) Alpha2beta1 integrin promotes chemoresistance against doxorubicin in cancer cells through extracellular signal-regulated kinase (ERK). J Biol Chem 287(21):17065–17076. doi:10.1074/jbc.M112.349365
Lee AJ, Endesfelder D, Rowan AJ, Walther A, Birkbak NJ, Futreal PA, Downward J, Szallasi Z, Tomlinson IP, Howell M, Kschischo M, Swanton C (2011) Chromosomal instability confers intrinsic multidrug resistance. Cancer Res 71(5):1858–1870. doi:10.1158/0008-5472.CAN-10-3604
Korner C, Keklikoglou I, Bender C, Worner A, Munstermann E, Wiemann S (2013) MicroRNA-31 sensitizes human breast cells to apoptosis by direct targeting of protein kinase C epsilon (PKCepsilon). J Biol Chem 288(12):8750–8761. doi:10.1074/jbc.M112.414128
Yahya SM, Elsayed GH (2014) A summary for molecular regulations of miRNAs in breast cancer. Clin Biochem. doi:10.1016/j.clinbiochem.2014.12.013
Farazi TA, Spitzer JI, Morozov P, Tuschl T (2011) miRNAs in human cancer. J Pathol 223(2):102–115. doi:10.1002/path.2806
Greve TS, Judson RL, Blelloch R (2013) microRNA control of mouse and human pluripotent stem cell behavior. Annu Rev Cell Dev Biol 29:213–239. doi:10.1146/annurev-cellbio-101512-122343
Zhu Y, Yu F, Jiao Y, Feng J, Tang W, Yao H, Gong C, Chen J, Su F, Zhang Y, Song E (2011) Reduced miR-128 in breast tumor-initiating cells induces chemotherapeutic resistance via Bmi-1 and ABCC5. Clin Cancer Res 17(22):7105–7115
Creevey L, Ryan J, Harvey H, Bray IM, Meehan M, Khan AR, Stallings RL (2013) MicroRNA-497 increases apoptosis in MYCN amplified neuroblastoma cells by targeting the key cell cycle regulator WEE1. Mol Cancer 12:23. doi:10.1186/1476-4598-12-23
Chen DQ, Pan BZ, Huang JY, Zhang K, Cui SY, De W, Wang R, Chen LB (2014) HDAC 1/4-mediated silencing of microRNA-200b promotes chemoresistance in human lung adenocarcinoma cells. Oncotarget 5(10):3333–3349
He X, Dong Y, Wu CW, Zhao Z, Ng SS, Chan FK, Sung JJ, Yu J (2012) MicroRNA-218 inhibits cell cycle progression and promotes apoptosis in colon cancer by downregulating BMI1 polycomb ring finger oncogene. Mol Med 18:1491–1498. doi:10.2119/molmed.2012.00304
Li J, Ping Z, Ning H (2012) miR-218 impairs tumor growth and increases chemo-sensitivity to cisplatin in cervical cancer. Int J Mol Sci 13(12):16053–16064. doi:10.3390/ijms131216053
Uesugi A, Kozaki K, Tsuruta T, Furuta M, Morita K, Imoto I, Omura K, Inazawa J (2011) The tumor suppressive microRNA miR-218 targets the mTOR component Rictor and inhibits AKT phosphorylation in oral cancer. Cancer Res 71(17):5765–5778. doi:10.1158/0008-5472.CAN-11-0368
Xin SY, Feng XS, Zhou LQ, Sun JJ, Gao XL, Yao GL (2014) Reduced expression of circulating microRNA-218 in gastric cancer and correlation with tumor invasion and prognosis. World J Gastroenterol 20(22):6906–6911. doi:10.3748/wjg.v20.i22.6906
Li Q, Zhu F, Chen P (2012) miR-7 and miR-218 epigenetically control tumor suppressor genes RASSF1A and Claudin-6 by targeting HoxB3 in breast cancer. Biochem Biophys Res Commun 424(1):28–33. doi:10.1016/j.bbrc.2012.06.028
He X, Xiao X, Dong L, Wan N, Zhou Z, Deng H, Zhang X (2014) miR-218 regulates cisplatin chemosensitivity in breast cancer by targeting BRCA1. Tumour Biol. doi:10.1007/s13277-014-2814-z
Raguz S, Adams C, Masrour N, Rasul S, Papoutsoglou P, Hu Y, Cazzanelli G, Zhou Y, Patel N, Coombes C, Yague E (2013) Loss of O(6)-methylguanine-DNA methyltransferase confers collateral sensitivity to carmustine in topoisomerase II-mediated doxorubicin resistant triple negative breast cancer cells. Biochem Pharmacol 85(2):186–196. doi:10.1016/j.bcp.2012.10.020
Zhou Y, Hu Y, Yang M, Jat P, Li K, Lombardo Y, Xiong D, Coombes RC, Raguz S, Yague E (2014) The miR-106b~25 cluster promotes bypass of doxorubicin-induced senescence and increase in motility and invasion by targeting the E-cadherin transcriptional activator EP300. Cell Death Differ 21(3):462–474. doi:10.1038/cdd.2013.167
Hu Y, Li S, Yang M, Yan C, Fan D, Zhou Y, Zhang Y, Yague E, Xiong D (2014) Sorcin silencing inhibits epithelial-to-mesenchymal transition and suppresses breast cancer metastasis in vivo. Breast Cancer Res Treat 143(2):287–299. doi:10.1007/s10549-013-2809-2
Hu Y, Cheng X, Li S, Zhou Y, Wang J, Cheng T, Yang M, Xiong D (2013) Inhibition of sorcin reverses multidrug resistance of K562/A02 cells and MCF-7/A02 cells via regulating apoptosis-related proteins. Cancer Chemother Pharmacol 72(4):789–798
Kogo R, How C, Chaudary N, Bruce J, Shi W, Hill RP, Zahedi P, Yip KW, Liu FF (2015) The microRNA-218~survivin axis regulates migration, invasion, and lymph node metastasis in cervical cancer. Oncotarget 6(2):1090–1100
Alajez NM, Lenarduzzi M, Ito E, Hui AB, Shi W, Bruce J, Yue S, Huang SH, Xu W, Waldron J, O’Sullivan B, Liu FF (2011) miR-218 suppresses nasopharyngeal cancer progression through downregulation of survivin and the SLIT2-ROBO1 pathway. Cancer Res 71(6):2381–2391. doi:10.1158/0008-5472.CAN-10-2754
Marusawa H, Matsuzawa S, Welsh K, Zou H, Armstrong R, Tamm I, Reed JC (2003) HBXIP functions as a cofactor of survivin in apoptosis suppression. EMBO J 22(11):2729–2740. doi:10.1093/emboj/cdg263
Dohi T, Okada K, Xia F, Wilford CE, Samuel T, Welsh K, Marusawa H, Zou H, Armstrong R, Matsuzawa S, Salvesen GS, Reed JC, Altieri DC (2004) An IAP-IAP complex inhibits apoptosis. J Biol Chem 279(33):34087–34090. doi:10.1074/jbc.C400236200
Wang S, Huang X, Lee CK, Liu B (2010) Elevated expression of erbB3 confers paclitaxel resistance in erbB2-overexpressing breast cancer cells via upregulation of Survivin. Oncogene 29(29):4225–4236. doi:10.1038/onc.2010.180
Zaffaroni N, Daidone MG (2002) Survivin expression and resistance to anticancer treatments: perspectives for new therapeutic interventions. Drug Res Updat 5(2):65–72
Pennati M, Folini M, Zaffaroni N (2008) Targeting survivin in cancer therapy. Exp Opin Ther Targets 12(4):463–476. doi:10.1517/14728222.12.4.463
Greinacher A, Selleng K (2010) Thrombocytopenia in the intensive care unit patient. Am Soc Hematol Edu Prog 2010:135–143. doi:10.1182/asheducation-2010.1.135
Baytekin F, Tuna B, Mungan U, Aslan G, Yorukoglu K (2011) Significance of P-glycoprotein, p53, and survivin expression in renal cell carcinoma. Urologic oncology 29(5):502–507. doi:10.1016/j.urolonc.2009.09.001
Song Z, Yao X, Wu M (2003) Direct interaction between survivin and Smac/DIABLO is essential for the anti-apoptotic activity of survivin during taxol-induced apoptosis. J Biol Chem 278(25):23130–23140. doi:10.1074/jbc.M300957200
Miller TE, Ghoshal K, Ramaswamy B, Roy S, Datta J, Shapiro CL, Jacob S, Majumder S (2008) MicroRNA-221/222 confers tamoxifen resistance in breast cancer by targeting p27Kip1. J Biol Chem 283(44):29897–29903. doi:10.1074/jbc.M804612200
Wang F, Li T, Zhang B, Li H, Wu Q, Yang L, Nie Y, Wu K, Shi Y, Fan D (2013) MicroRNA-19a/b regulates multidrug resistance in human gastric cancer cells by targeting PTEN. Biochem Biophys Res Commun 434(3):688–694. doi:10.1016/j.bbrc.2013.04.010
Liang Z, Li Y, Huang K, Wagar N, Shim H (2011) Regulation of miR-19 to breast cancer chemoresistance through targeting PTEN. Pharm Res 28(12):3091–3100. doi:10.1007/s11095-011-0570-y
Liu J, Chen G, Feng L, Zhang W, Pelicano H, Wang F, Ogasawara MA, Lu W, Amin HM, Croce CM, Keating MJ, Huang P (2014) Loss of p53 and altered miR15-a/16-1short right arrowMCL-1 pathway in CLL: insights from TCL1-Tg:p53(-/-) mouse model and primary human leukemia cells. Leukemia 28(1):118–128. doi:10.1038/leu.2013.125
Cai CK, Zhao GY, Tian LY, Liu L, Yan K, Ma YL, Ji ZW, Li XX, Han K, Gao J, Qiu XC, Fan QY, Yang TT, Ma BA (2012) miR-15a and miR-16-1 downregulate CCND1 and induce apoptosis and cell cycle arrest in osteosarcoma. Oncol Rep 28(5):1764–1770. doi:10.3892/or.2012.1995
Martinez I, Gardiner AS, Board KF, Monzon FA, Edwards RP, Khan SA (2008) Human papillomavirus type 16 reduces the expression of microRNA-218 in cervical carcinoma cells. Oncogene 27(18):2575–2582. doi:10.1038/sj.onc.1210919
Lowery AJ, Miller N, Devaney A, McNeill RE, Davoren PA, Lemetre C, Benes V, Schmidt S, Blake J, Ball G, Kerin MJ (2009) MicroRNA signatures predict oestrogen receptor, progesterone receptor and HER2/neu receptor status in breast cancer. Breast Cancer Res 11(3):R27. doi:10.1186/bcr2257
Zhang XL, Shi HJ, Wang JP, Tang HS, Wu YB, Fang ZY, Cui SZ, Wang LT (2014) MicroRNA-218 is upregulated in gastric cancer after cytoreductive surgery and hyperthermic intraperitoneal chemotherapy and increases chemosensitivity to cisplatin. World J Gastroenterol 20(32):11347–11355. doi:10.3748/wjg.v20.i32.11347
Li F, Ambrosini G, Chu EY, Plescia J, Tognin S, Marchisio PC, Altieri DC (1998) Control of apoptosis and mitotic spindle checkpoint by survivin. Nature 396(6711):580–584. doi:10.1038/25141
Altieri DC (2008) Survivin, cancer networks and pathway-directed drug discovery. Nat Rev Cancer 8(1):61–70. doi:10.1038/nrc2293
Kanwar JR, Kamalapuram SK, Kanwar RK (2013) Survivin signaling in clinical oncology: a multifaceted dragon. Med Res Rev 33(4):765–789. doi:10.1002/med.21264
Singh N, Krishnakumar S, Kanwar RK, Cheung CH, Kanwar JR (2014) Clinical aspects for survivin: a crucial molecule for targeting drug-resistant cancers. Drug Discov Today. doi:10.1016/j.drudis.2014.11.013
Nabilsi NH, Broaddus RR, Loose DS (2009) DNA methylation inhibits p53-mediated survivin repression. Oncogene 28(19):2046–2050. doi:10.1038/onc.2009.62
Huang J, Lyu H, Wang J, Liu B (2015) MicroRNA regulation and therapeutic targeting of survivin in cancer. Am J Cancer Res 5(1):20–31
Wu DW, Cheng YW, Wang J, Chen CY, Lee H (2010) Paxillin predicts survival and relapse in non-small cell lung cancer by microRNA-218 targeting. Cancer Res 70(24):10392–10401. doi:10.1158/0008-5472.CAN-10-2341
Nakahara T, Kita A, Yamanaka K, Mori M, Amino N, Takeuchi M, Tominaga F, Hatakeyama S, Kinoyama I, Matsuhisa A, Kudoh M, Sasamata M (2007) YM155, a novel small-molecule survivin suppressant, induces regression of established human hormone-refractory prostate tumor xenografts. Cancer Res 67(17):8014–8021. doi:10.1158/0008-5472.CAN-07-1343
Holmes D (2012) Cancer drug’s survivin suppression called into question. Nat Med 18(6):842–843. doi:10.1038/nm0612-842b
Betel D, Wilson M, Gabow A, Marks DS, Sander C (2008) The microRNA.org resource: targets and expression. Nucleic Acids Res 36 (Database issue):D149–153. doi:10.1093/nar/gkm995
Acknowledgments
We thank Prof. Dongsheng Xiong (Institute of Hematology, PUMC, Tianjin, China) for a generous gift of human breast cancer MCF-7 and MCF-7/A02 cells. This work was supported by the Chinese National Natural Sciences Foundation (81402480 to YH), the Science and Technology Foundation of Tianjin Municipal Health Bureau (2014KZ078 to YH) and the China Scholarship Council (to YH) for visiting Imperial College London.
Conflict of interest
The authors declared no conflict of interest.
Ethical approval
All procedures performed in studies involving animals performed at the National Institutes of Health (Tianjin Cancer Hospital) in accordance with guidelines under the Institutional Animal Care and Use Committee (IACUC) and approved by the Committee on the Ethics of Animal Experiments of the Tianjin Cancer Hospital. All applicable national and institutional guidelines for the care and use of animals were followed.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Yunhui Hu, Kun Xu, and Ernesto Yagüe contributed equally to this study.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Hu, Y., Xu, K. & Yagüe, E. miR-218 targets survivin and regulates resistance to chemotherapeutics in breast cancer. Breast Cancer Res Treat 151, 269–280 (2015). https://doi.org/10.1007/s10549-015-3372-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-015-3372-9