Abstract
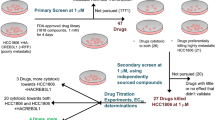
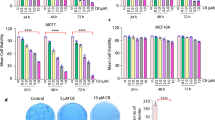
The treatment of patients with invasive breast cancer remains a major issue because of the acquisition of drug resistance to conventional chemotherapy. Here we propose a new therapeutic strategy by combining DNA methyltransferase inhibitors (DMTIs) with suramin. Cytotoxic effects of suramin or combination treatment with DMTIs were determined in highly invasive breast cancer cell lines MDA-MB-231, BT-20 and HCC1954, or control cells. In addition, effects on cell invasion were determined in 3-dimensional cell culture assays. DMTI-mediated upregulation of Protein Kinase D1 (PKD1) expression was shown by Western blotting. Effects of suramin on PKD1 activity was determined in vitro and in cells. The importance of PKD1 in mediating the effects of such combination treatment in cell invasion was demonstrated using 3D cell culture assays. A proof of principal animal experiment was performed showing that PKD1 is critical for breast cancer growth. We show that when used in combination, suramin and DMTIs impair the invasive phenotype of breast cancer cells. We show that PKD1, a kinase that previously has been described as a suppressor of tumor cell invasion, is an interface for both FDA-approved drugs, since the additive effects observed are due to DMTI-mediated re-expression and suramin-induced activation of PKD1. Our data reveal a mechanism of how a combination treatment with non-toxic doses of suramin and DMTIs may be of therapeutic benefit for patients with aggressive, multi-drug resistant breast cancer.






Similar content being viewed by others
Abbreviations
- BC:
-
Breast cancer
- DMTI:
-
DNA methyltransferase inhibitor
- EMT:
-
Epithelial-to-mesenchymal transition
- FFPE:
-
Formalin-fixed paraffin-embedded
- GF:
-
Growth factor
- IHC:
-
Immunohistochemistry
- Mfp:
-
Mammary fat pad
- MMP:
-
Matrix metalloproteinase
- PKC:
-
Protein kinase C
- PKD:
-
Protein kinase D
- RT:
-
Room temperature
- TN:
-
Triple negative
References
Karahoca M, Momparler RL (2013) Pharmacokinetic and pharmacodynamic analysis of 5-aza-2′-deoxycytidine (decitabine) in the design of its dose-schedule for cancer therapy. Clin Epigenet 5:3
Singh V, Sharma P, Capalash N (2013) DNA methyltransferase inhibitors as epigenetic therapy for cancer. Curr Cancer Drug Targets 13:379–399
Karpf AR, Moore BC, Ririe TO, Jones DA (2001) Activation of the p53 DNA damage response pathway after inhibition of DNA methyltransferase by 5-aza-2′-deoxycytidine. Mol Pharmacol 59:751–757
Eiseler T, Doppler H, Yan IK, Goodison S, Storz P (2009) Protein kinase D1 regulates matrix metalloproteinase expression and inhibits breast cancer cell invasion. Breast Cancer Res 11:R13
Borges S, Doppler H, Perez EA, Andorfer CA, Sun Z et al (2013) Pharmacologic reversion of epigenetic silencing of the PRKD1 promoter blocks breast tumor cell invasion and metastasis. Breast Cancer Res 15:R66
Chopin DK, Caruelle JP, Colombel M, Palcy S, Ravery V et al (1993) Increased immunodetection of acidic fibroblast growth factor in bladder cancer, detectable in urine. J Urol 150:1126–1130
Cronauer MV, Hittmair A, Eder IE, Hobisch A, Culig Z et al (1997) Basic fibroblast growth factor levels in cancer cells and in sera of patients suffering from proliferative disorders of the prostate. Prostate 31:223–233
Ravery V, Jouanneau J, Gil Diez S, Abbou CC, Caruelle JP et al (1992) Immunohistochemical detection of acidic fibroblast growth factor in bladder transitional cell carcinoma. Urol Res 20:211–214
Ropiquet F, Giri D, Kwabi-Addo B, Mansukhani A, Ittmann M (2000) Increased expression of fibroblast growth factor 6 in human prostatic intraepithelial neoplasia and prostate cancer. Cancer Res 60:4245–4250
Singh RK, Bucana CD, Gutman M, Fan D, Wilson MR et al (1994) Organ site-dependent expression of basic fibroblast growth factor in human renal cell carcinoma cells. Am J Pathol 145:365–374
Suzuki K, Tokue A, Kamiakito T, Kuriki K, Saito K et al (2001) Predominant expression of fibroblast growth factor (FGF) 8, FGF4, and FGF receptor 1 in nonseminomatous and highly proliferative components of testicular germ cell tumors. Virchows Arch 439:616–621
Bernsen HJ, Rijken PF, Peters JP, Bakker JH, Boerman RH et al (1999) Suramin treatment of human glioma xenografts; effects on tumor vasculature and oxygenation status. J Neurooncol 44:129–136
Bhargava S, Hotz B, Hines OJ, Reber HA, Buhr HJ et al (2007) Suramin inhibits not only tumor growth and metastasis but also angiogenesis in experimental pancreatic cancer. J Gastrointest Surg 11:171–178
Danesi R, Del Bianchi S, Soldani P, Campagni A, La Rocca RV et al (1993) Suramin inhibits bFGF-induced endothelial cell proliferation and angiogenesis in the chick chorioallantoic membrane. Br J Cancer 68:932–938
Hawking F (1978) Suramin: with special reference to onchocerciasis. Adv Pharmacol Chemother 15:289–322
Coffey RJ Jr, Goustin AS, Soderquist AM, Shipley GD, Wolfshohl J et al (1987) Transforming growth factor alpha and beta expression in human colon cancer lines: implications for an autocrine model. Cancer Res 47:4590–4594
Hosang M (1985) Suramin binds to platelet-derived growth factor and inhibits its biological activity. J Cell Biochem 29:265–273
Pollak M, Richard M (1990) Suramin blockade of insulinlike growth factor I-stimulated proliferation of human osteosarcoma cells. J Natl Cancer Inst 82:1349–1352
Williams LT, Tremble PM, Lavin MF, Sunday ME (1984) Platelet-derived growth factor receptors form a high affinity state in membrane preparations. Kinetics and affinity cross-linking studies. J Biol Chem 259:5287–5294
Hensey CE, Boscoboinik D, Azzi A (1989) Suramin, an anti-cancer drug, inhibits protein kinase C and induces differentiation in neuroblastoma cell clone NB2A. FEBS Lett 258:156–158
Buchinger B, Spitzer S, Karlic H, Klaushofer K, Varga F (2008) Lysyl oxidase (LOX) mRNA expression and genes of the differentiated osteoblastic phenotype are upregulated in human osteosarcoma cells by suramin. Cancer Lett 265:45–54
Wiese C, Nikolova T, Zahanich I, Sulzbacher S, Fuchs J et al (2011) Differentiation induction of mouse embryonic stem cells into sinus node-like cells by suramin. Int J Cardiol 147:95–111
Jiang S, Chen X, Li C, Zhang X, Zhang T et al (2012) Suramin inhibits the growth of nasopharyngeal carcinoma cells via the downregulation of osteopontin. Mol Med Rep 6:1351–1354
Stein CA (1993) Suramin: a novel antineoplastic agent with multiple potential mechanisms of action. Cancer Res 53:2239–2248
Bastea LI, Doppler H, Balogun B, Storz P (2012) Protein kinase D1 maintains the epithelial phenotype by inducing a DNA-bound, inactive SNAI1 transcriptional repressor complex. PLoS ONE 7:e30459
Du C, Zhang C, Hassan S, Biswas MH, Balaji KC (2010) Protein kinase D1 suppresses epithelial-to-mesenchymal transition through phosphorylation of snail. Cancer Res 70:7810–7819
Jaggi M, Rao PS, Smith DJ, Hemstreet GP, Balaji KC (2003) Protein kinase C mu is down-regulated in androgen-independent prostate cancer. Biochem Biophys Res Commun 307:254–260
Olayioye MA, Barisic S, Hausser A (2013) Multi-level control of actin dynamics by protein kinase D. Cell Signal 25:1739–1747
Storz P, Doppler H, Johannes FJ, Toker A (2003) Tyrosine phosphorylation of protein kinase D in the pleckstrin homology domain leads to activation. J Biol Chem 278:17969–17976
Cowell CF, Doppler H, Yan IK, Hausser A, Umezawa Y et al (2009) Mitochondrial diacylglycerol initiates protein-kinase D1-mediated ROS signaling. J Cell Sci 122:919–928
Guo H, Liu W, Ju Z, Tamboli P, Jonasch E et al (2012) An efficient procedure for protein extraction from formalin-fixed, paraffin-embedded tissues for reverse phase protein arrays. Proteome Sci 10:56
Vignon F, Prebois C, Rochefort H (1992) Inhibition of breast cancer growth by suramin. J Natl Cancer Inst 84:38–42
Song S, Yu B, Wei Y, Wientjes MG, Au JL (2004) Low-dose suramin enhanced paclitaxel activity in chemotherapy-naive and paclitaxel-pretreated human breast xenograft tumors. Clin Cancer Res 10:6058–6065
Waldron RT, Rozengurt E (2003) Protein kinase C phosphorylates protein kinase D activation loop Ser744 and Ser748 and releases autoinhibition by the pleckstrin homology domain. J Biol Chem 278:154–163
Gschwendt M, Kittstein W, Johannes FJ (1998) Differential effects of suramin on protein kinase C isoenzymes. A novel tool for discriminating protein kinase C activities. FEBS Lett 421:165–168
Matthews SA, Rozengurt E, Cantrell D (1999) Characterization of serine 916 as an in vivo autophosphorylation site for protein kinase D/Protein kinase Cmu. J Biol Chem 274:26543–26549
Storz P, Doppler H, Copland JA, Simpson KJ, Toker A (2009) FOXO3a promotes tumor cell invasion through the induction of matrix metalloproteinases. Mol Cell Biol 29:4906–4917
Bowden CJ, Figg WD, Dawson NA, Sartor O, Bitton RJ et al (1996) A phase I/II study of continuous infusion suramin in patients with hormone-refractory prostate cancer: toxicity and response. Cancer Chemother Pharmacol 39:1–8
Dreicer R, Smith DC, Williams RD, See WA (1999) Phase II trial of suramin in patients with metastatic renal cell carcinoma. Invest New Drugs 17:183–186
Falcone A, Pfanner E, Cianci C, Danesi R, Brunetti I et al (1995) Suramin in patients with metastatic colorectal cancer pretreated with fluoropyrimidine-based chemotherapy. A phase II study. Cancer 75:440–443
George S, Dreicer R, Au JJ, Shen T, Rini BI et al (2008) Phase I/II trial of 5-fluorouracil and a noncytotoxic dose level of suramin in patients with metastatic renal cell carcinoma. Clin Genitourin Cancer 6:79–85
Lustberg MB, Pant S, Ruppert AS, Shen T, Wei Y et al (2012) Phase I/II trial of non-cytotoxic suramin in combination with weekly paclitaxel in metastatic breast cancer treated with prior taxanes. Cancer Chemother Pharmacol 70:49–56
Villalona-Calero MA, Otterson GA, Wientjes MG, Weber F, Bekaii-Saab T et al (2008) Noncytotoxic suramin as a chemosensitizer in patients with advanced non-small-cell lung cancer: a phase II study. Ann Oncol 19:1903–1909
Villalona-Calero MA, Wientjes MG, Otterson GA, Kanter S, Young D et al (2003) Phase I study of low-dose suramin as a chemosensitizer in patients with advanced non-small cell lung cancer. Clin Cancer Res 9:3303–3311
Song S, Wientjes MG, Gan Y, Au JL (2000) Fibroblast growth factors: an epigenetic mechanism of broad spectrum resistance to anticancer drugs. Proc Natl Acad Sci U S A 97:8658–8663
Song S, Wientjes MG, Walsh C, Au JL (2001) Nontoxic doses of suramin enhance activity of paclitaxel against lung metastases. Cancer Res 61:6145–6150
Zhang Y, Song S, Yang F, Au JL, Wientjes MG (2001) Nontoxic doses of suramin enhance activity of doxorubicin in prostate tumors. J Pharmacol Exp Ther 299:426–433
Skliris GP, Munot K, Bell SM, Carder PJ, Lane S et al (2003) Reduced expression of oestrogen receptor beta in invasive breast cancer and its re-expression using DNA methyl transferase inhibitors in a cell line model. J Pathol 201:213–220
Zhu WG, Hileman T, Ke Y, Wang P, Lu S et al (2004) 5-Aza-2′-deoxycytidine activates the p53/p21Waf1/Cip1 pathway to inhibit cell proliferation. J Biol Chem 279:15161–15166
Constantinides PG, Jones PA, Gevers W (1977) Functional striated muscle cells from non-myoblast precursors following 5-azacytidine treatment. Nature 267:364–366
Jones PA, Taylor SM (1980) Cellular differentiation, cytidine analogs and DNA methylation. Cell 20:85–93
Eiseler T, Doppler H, Yan IK, Kitatani K, Mizuno K et al (2009) Protein kinase D1 regulates cofilin-mediated F-actin reorganization and cell motility through slingshot. Nat Cell Biol 11:545–556
Eiseler T, Hausser A, De Kimpe L, Van Lint J, Pfizenmaier K (2010) Protein kinase D controls actin polymerization and cell motility through phosphorylation of cortactin. J Biol Chem 285:18672–18683
Eiseler T, Schmid MA, Topbas F, Pfizenmaier K, Hausser A (2007) PKD is recruited to sites of actin remodelling at the leading edge and negatively regulates cell migration. FEBS Lett 581:4279–4287
Peterburs P, Heering J, Link G, Pfizenmaier K, Olayioye MA et al (2009) Protein kinase D regulates cell migration by direct phosphorylation of the cofilin phosphatase slingshot 1 like. Cancer Res 69:5634–5638
Spratley SJ, Bastea LI, Doppler H, Mizuno K, Storz P (2011) Protein kinase D regulates cofilin activity through p21-activated kinase 4. J Biol Chem 286:34254–34261
Kim M, Jang HR, Kim JH, Noh SM, Song KS et al (2008) Epigenetic inactivation of protein kinase D1 in gastric cancer and its role in gastric cancer cell migration and invasion. Carcinogenesis 29:629–637
Onishi Y, Kawamoto T, Kishimoto K, Hara H, Fukase N et al (2012) PKD1 negatively regulates cell invasion, migration and proliferation ability of human osteosarcoma. Int J Oncol 40:1839–1848
Isham CR, Tibodeau JD, Jin W, Xu R, Timm MM et al (2007) Chaetocin: a promising new antimyeloma agent with in vitro and in vivo activity mediated via imposition of oxidative stress. Blood 109:2579–2588
Sundram V, Chauhan SC, Ebeling M, Jaggi M (2012) Curcumin attenuates beta-catenin signaling in prostate cancer cells through activation of protein kinase D1. PLoS ONE 7:e35368
Acknowledgments
This work was supported by grants from the NIH (GM086435) and the Bankhead-Coley Program of the Florida Department of Health (1BG11). Research reported in this publication was also supported by the National Cancer Institute of the National Institutes of Health under award number P50CA116201. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Cancer Institute or the National Institutes of Health. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript. We also thank the Luther and Susie Harrison Foundation for their support, Irene K. Yan for technical assistance, and Alicia Fleming for help with the manuscript.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Borges, S., Döppler, H.R. & Storz, P. A combination treatment with DNA methyltransferase inhibitors and suramin decreases invasiveness of breast cancer cells. Breast Cancer Res Treat 144, 79–91 (2014). https://doi.org/10.1007/s10549-014-2857-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-014-2857-2