Abstract
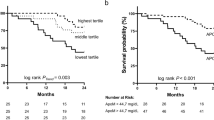
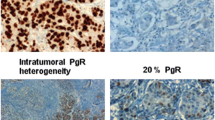
Apolipoprotein D (ApoD) is a lipocalin involved in various cellular processes, including cytoprotection. We retrospectively evaluated its prognostic impact in 272 women (median age 63 (range 21–89) years, with operable breast cancer (Stage I or IIa) treated according to national guidelines, stratified by age and menopausal status, with a median long-term follow-up of 12 years. ApoD, estrogen receptor-alpha (ERα), and progesterone receptor (PR) positivity were determined by quantitative immunohistochemistry in tissue microarrays taken from the invasive front of the tumors and in whole tumor sections. ApoD was expressed in 50% of the tumors. Diffuse nuclear and cytoplasmic (ApoDCN), but not granular cytoplasmic ApoD expression in the invasive front was a detrimental factor for elderly patients over 70 years of age, (n = 83), but not for younger and pre-menopausal patients. ApoDCN expression correlated with lymph node metastases (P = 0.04). Moreover, a correlation between ApoDCN localization and age over 70 years was observed (P = 0.03). In the elderly, ApoDCN positive tumors had both a significantly shorter relapse-free survival (all locations) (P = 0.02) and a decreased breast cancer-specific survival (P < 0.0001). Moreover, the prognostic importance of ApoD was dose dependent (P = 0.002) Multivariate analysis confirmed the prognostic value of ApoDCN. When the elderly patients were stratified by nodal status, ApoD showed prognostic importance only in the node positive patients (n = 30) (HR = 9.6; 95% CI = 2.3–40.4). The potential relevance of ApoD in this increasingly frequent group of breast cancer patients should be further explored.




Similar content being viewed by others
References
Veronesi U, Boyle P, Goldhirsch A et al (2005) Breast cancer. Lancet 365:1727–1741
Walker RA (2003) Prognostic and predictive factors in breast cancer: an overview. In: Walker RA (ed) Prognostic and predictive factors in breast cancer, Martin Duniz, Taylor & Francis, UK, pp 1–5
Søreide JA, Lea OA, Kvinnsland S (1987) Progesterone binding cyst protein (PBCP) in primary breast cancer: a new prognostic factor? Breast Cancer Res Treat 9:123–128
Søreide JA, Lea OA, Anda O et al (1991) Progesterone-binding cyst protein (PBCP) in operable breast cancer: correlations with prognostic factors and predictive value for effect of adjuvant tamoxifen treatment. Anticancer Res 11:601–605
Diez-Itza I, Vizoso F, Merino AM et al (1994) Expression and prognostic significance of apolipoprotein D in breast cancer. Am J Pathol 144:310–320
Flower DR (2000) The lipocalin protein family: structural and sequence overview. Biochem Biophys Acta 1482:9–24
Ayrault Jarrier M, Levy G, Polonovski J (1963) Study of humanserum alpha-lipoproteins by immunoelectrophoresis. Bull Soc Chim Biol (Paris) 45:703–713
Pearlman WH, Gueriguian JL, Sawyer ME (1973) A specific progesterone-binding component of human breast cyst fluid. J Biol Chem 248:5736–5741
Haagensen DE Jr, Mazoujian G, Holder WD Jr et al (1977) Evaluation of a breast cyst fluid protein detectable in the plasma of breast carcinoma patients. Ann Surg 185:279–285
Lea OA, Kvinnsland S, Thorsen T (1987) Progesterone-binding cyst protein in human breast tumor cytosol. Cancer Res 47:6189–6192
Balbin M, Freije JM, Fueyo A et al (1990) Apolipoprotein D is the major protein component in cyst fluid from women with human breast gross cystic disease. Biochem J 271:803–807
Rassart E, Bedirian A, Do Carmo S et al (2000) Apolipoprotein D. Biochim Biophys Acta 1482:185–198
Online Mendelinan Inheritance in Man™ (OMIM™), McKusick-Nathans Institute of Genetic Medicine, Johns Hopkins University (Baltimore, MD) and National Center for Biotechnology Information, National Library of Medicine (Bethesda, MD), World Wide Web URL:/http://www.ncbi.nlm.nih.gov/entrez/dispomim.cgi?id=107740. Cited February 7, 2008
Morais Cabral JH, Atkins GL, Sanchez LM et al (1995) Arachidonic acid binds to apolipoprotein D: implications for the protein's function. FEBS Lett 366:53–56
Romano M, Claria J (2003) Cyclooxygenase-2 and 5-lipoxygenase converging functions on cell proliferation and tumor angiogenesis: implications for cancer therapy. FASEB J 17:1986–1995
Simard J, de Launoit Y, Haagensen DE et al (1992) Additive stimulatory action of glucocorticoids and androgens on basal and estrogen-repressed apolipoprotein-D messenger ribonucleic acid levels and secretion in human breast cancer cells. Endocrinology 130:1115–1121
Lea OA (1988) Binding properties of progesterone-binding Cyst protein, PBCP. Steroids 52:337–338
Harding C, Osundeko O, Tetlow L et al (2000) Hormonally-regulated proteins in breast secretions are markers of target organ sensitivity. Br J Cancer 82:354–360
Do Carmo S, Levros LC Jr, Rassart E (2007) Modulation of apolipoprotein D expression and translocation under specific stress conditions. Biochim Biophys Acta 1773:954–969
Thomas EA, Copolov DL, Sutcliffe JG (2003) From pharmacotherapy to pathophysiology: emerging mechanisms of apolipoprotein D in psychiatric disorders. Curr Mol Med 3:408–418
West RB, Nuyten DS, Subramanian S et al (2005) Determination of stromal signatures in breast carcinoma. PLoS Biol 3:e187
Søiland H, Søreide K, Janssen EAM et al (2007) Emerging concepts of apolipoprotein D with possible implications for breast cancer. Cell Oncol 29:195–209
Søiland H, Skaland I, Janssen EAM et al (2008) Comparison of Apolipoprotein D determination methods in breast cancer. Anticancer Res 28:673–680
Søreide JA, Lea OA, Varhaug JE et al (1992) Androgen receptors in operable breast cancer: relation to other steroid hormone receptors, correlations to prognostic factors and predictive value for effect of adjuvant tamoxifen treatment. Eur J Surg Oncol 18:112–118
Sobin LH, Witterkind C (2002) Breast tumors. In: TNM classifications of malignant tumors, 6th edn. International Union Against Cancer (UICC). John Wiley & Sons, Hoboken, New Jersey, pp 131–142
Tavassoli FA, Devilee P (2003) Pathology and genetics of tumours of the breast and female genital organs. In: World Health Organization classification of tumours. IARC Press, Lyon, pp 9–112
Baak JP, van Diest PJ, Voorhorst FJ et al (2007) The prognostic value of proliferation in lymph-node-negative breast cancer patients is age dependent. Eur J Cancer 43:527–535
Crivellari D, Aapro M, Leonard R et al (2007) Breast cancer in the elderly. J Clin Oncol 25:1882–1890
Baak JP, van Diest PJ, Voorhorst FJ et al (2005) Prospective multicenter validation of the independent prognostic value of the mitotic activity index in lymph node-negative breast cancer patients younger than 55 years. J Clin Oncol 23:5993–6001
Kallioniemi OP, Wagner U, Kononen J et al (2001) Tissue microarray technology for high-throughput molecular profiling of cancer. Hum Mol Genet 10:657–662
Henriksen KL, Rasmussen BB, Lykkesfeldt AE et al (2007) Semi-quantitative scoring of potentially predictive markers for endocrine treatment of breast cancer: a comparison between whole sections and tissue microarrays. J Clin Pathol 60:397–404
Janssen EA, van Diest PJ, Søiland H et al (2006) Success predictors of adjuvant chemotherapy in node-negative breast cancer patients under 55 years. Cell Oncol 28:295–303
Baker SG (2003) The central role of receiver operating characteristic (ROC) curves in evaluating tests for the early detection of cancer. J Natl Cancer Inst 95:511–515
Zlobec I, Steele R, Terracciano L et al (2007) Selecting immunohistochemical cut-off scores for novel biomarkers of progression and survival in colorectal cancer. J Clin Pathol 60:1112–1116
Zweig MH, Campbell G (1993) Receiver-operating characteristic (ROC) plots: a fundamental evaluation tool in clinical medicine. Clin Chem 39:561–577
Altman DG, Lausen B, Sauerbrei W et al (1994) Dangers of using "optimal" cutpoints in the evaluation of prognostic factors. J Natl Cancer Inst 86:829–835
Hermann RE, Steiger E (1978) Modified radical mastectomy. Surg Clin North Am 58:743–754
The Norwegian Cancer Society, The Norwegian Surgical Society, The Norwegian Society of Therapeutic Oncology (1981) Primary treatment of breast cancer (National guidelines) (in Norwegian)
Gundersen S, Hannisdal E, Søreide JA et al (1995) Adjuvant tamoxifen for pre- and postmenopausal women with estrogen receptor positive, node positive breast cancer: a randomized study. Breast Cancer Res Treat 36:49–53
Organization web site: Norwegian Breast Cancer Group, http://www.nbcg.net. Cited 5 Jan 2008
Lamelas ML, Vazquez J, Enguita MI et al (2000) Apolipoprotein D expression in metastasic lymph nodes of breast cancer. Int J Surg Investig 2:285–293
Carreno G, Del Casar JM, Corte MD et al (2007) Local recurrence after mastectomy for breast cancer: analysis of clinicopathological, biological and prognostic characteristics. Breast Cancer Res Treat 102:61–73
Torhorst J, Bucher C, Kononen J et al (2001) Tissue microarrays for rapid linking of molecular changes to clinical endpoints. Am J Pathol 159:2249–2256
Polkowski W, Meijer GA, Baak JP et al (1997) Reproducibility of p53 and Ki-67 immunoquantitation in Barrett's esophagus. Anal Quant Cytol Histol 19:246–254
Gennari R, Curigliano G, Rotmensz N et al (2004) Breast carcinoma in elderly women: features of disease presentation, choice of local and systemic treatments compared with younger postmenopausal patients. Cancer 101:1302–1310
Muss HB, Biganzoli L, Sargent DJ et al (2007) Adjuvant therapy in the elderly: making the right decision. J Clin Oncol 25:1870–1875
Aspinall JO, Bentel JM, Horsfall DJ et al (1995) Differential expression of apolipoprotein-D and prostate specific antigen in benign and malignant prostate tissues. J Urol 154:622–628
Bianchi-Frias D, Pritchard C, Mecham BH et al (2007) Genetic background influences murine prostate gene expression: implications for cancer phenotypes. Genome Biol 8:R117
Hall RE, Horsfall DJ, Stahl J et al (2004) Apolipoprotein-D: a novel cellular marker for HGPIN and prostate cancer. Prostate 58:103–108
Miranda E, Vizoso F, Martin A et al (2003) Apolipoprotein D expression in cutaneous malignant melanoma. J Surg Oncol 83:99–105
Sanchez D, Lopez-Arias B, Torroja L et al (2006) Loss of glial lazarillo, a homolog of apolipoprotein D, reduces lifespan and stress resistance in Drosophila. Curr Biol 16:680–686
Walker DW, Muffat J, Rundel C et al (2006) Overexpression of a Drosophila homolog of apolipoprotein D leads to increased stress resistance and extended lifespan. Curr Biol 16:674–679
Janssen EA, Baak JP, Guervos MA et al (2003) In lymph node-negative invasive breast carcinomas, specific chromosomal aberrations are strongly associated with high mitotic activity and predict outcome more accurately than grade, tumour diameter, and oestrogen receptor. J Pathol 201:555–561
Skerra A (2007) Anticalins as alternative binding proteins for therapeutic use. Curr Opin Mol Ther 9:336–344
Acknowledgements
Dr. Håvard Søiland, MD, is a Research Fellow, financially sponsored by the Western Norwegian Health Authorities (project # 911166). Financial support from The Norwegian Cancer Society, The L. Meltzer Foundation, and O. Mjålands Foundation is highly appreciated. We are grateful for the technical assistance of Britt Fjæran.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Søiland, H., Janssen, E.A.M., Kørner, H. et al. Apolipoprotein D predicts adverse outcome in women ≥70 years with operable breast cancer. Breast Cancer Res Treat 113, 519–528 (2009). https://doi.org/10.1007/s10549-008-9955-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-008-9955-y