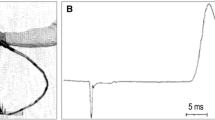
Abstract
Objective
To systematically review the literature on the use of the transcranial static magnetic stimulation (tSMS) technique in humans and animals, its effects on different areas of the central nervous system (CNS), its influence on neural excitability and on the subject’s behavior, and its biological effects and future possibilities. All static magnetic field applications that can be considered to have a physiologically similar effect have been reviewed.
Methods
We searched studies using key terms in NCBI PubMed, Scopus, PEDro, SciELO, Cochrane, and links to publications (inception to September 2019). Three reviewers independently selected the studies, extracted data, and assessed the methodological quality of the studies using the recommendations described in the Cochrane Handbook for Systematic Reviews of Interventions, PRISMA guidelines.
Results
We analyzed 27 studies. The reviewed literature suggests that the use of these magnetic fields has an inhibitory effect on different areas of the CNS, such as motor, somatosensory, and visual cortex, cerebellum, and spinal cord. Regarding subject’s behavior, the different effects of tSMS appear to be transient and dependent on the stimulated area, such as loss of visual discrimination or improvement of somatosensory perception. In addition, the technique has some therapeutic utility, specifically in pathologies with cortical hyperexcitability.
Conclusions
These results suggest that tSMS may be a promising tool to modulate cerebral excitability in a safe and non-invasive way. Further investigations could give a better explanation of its precise mechanisms of action and applications.


Similar content being viewed by others
References
Zhu H, Lu Z, Jin Y, Duan X, Teng J, Duan D (2015) Low-frequency repetitive transcranial magnetic stimulation on Parkinson motor function: a meta-analysis of randomised controlled trials. Acta Neuropsychiatr 27(02):82–89. https://doi.org/10.1017/neu.2014.43
Huang Y-Z, Edwards MJ, Rounis E, Bhatia KP, Rothwell JC (2005) Theta burst stimulation of the human motor cortex. Neuron 45(2):201–206. https://doi.org/10.1016/j.neuron.2004.12.033
Nitsche MA, Cohen LG, Wassermann EM et al (2008) Transcranial direct current stimulation: state of the art 2008. Brain Stimul 1(3):206–223. https://doi.org/10.1016/j.brs.2008.06.004
Oliviero A, Mordillo-Mateos L, Arias P, Panyavin I, Foffani G (2011) & Aguilar, Juan. Transcranial static magnetic field stimulation of the human motor cortex. J Physiol 589(20):4949–4958. https://doi.org/10.1113/jphysiol.2011.211953
Pascual-Leone A, Freitas C, Oberman L et al (2011) Characterizing brain cortical plasticity and network dynamics across the age-span in health and disease with TMS-EEG and TMS-fMRI. Brain Topogr 24(3-4):302–315. https://doi.org/10.1007/s10548-011-0196-8
Klomjai W, Katz R, Lackmy-Vallée A (2015) Basic principles of transcranial magnetic stimulation (TMS) and repetitive TMS (rTMS). Ann Phys Rehabil Med 58(4):208–213. https://doi.org/10.1016/j.rehab.2015.05.005
Paulus W (2011) Transcranial electrical stimulation (tES - tDCS; tRNS, tACS) methods. Neuropsychol Rehabil 21(5):602–617. https://doi.org/10.1080/09602011.2011.557292
Stagg CJ, Nitsche MA (2011) Physiological basis of transcranial direct current stimulation. Neuroscientist. 17(1):37–53. https://doi.org/10.1177/1073858410386614
Sánchez-León CA, Sánchez-López Á, Ammann C, Cordones I, Carretero-Guillén A, Márquez-Ruiz J (2018) Exploring new transcranial electrical stimulation strategies to modulate brain function in animal models. Curr Opin Biomed Eng 8:7–13. https://doi.org/10.1016/j.cobme.2018.09.001
Rosen AD (2003) Mechanism of action of moderate-intensity static magnetic fields on biological systems. Cell Biochem Biophys 39(2):163–174. https://doi.org/10.1385/CBB:39:2:163
Silbert BI, Pevcic DD, Patterson HI, Windnagel KA, Thickbroom GW (2003) Inverse correlation between resting motor threshold and corticomotor excitability after static magnetic stimulation of human motor cortex. Brain Stimul 6(5):817–820. https://doi.org/10.1016/j.brs.2013.03.007
Nojima I, Koganemaru S, Fukuyama H, Mima T (2015) Static magnetic field can transiently alter the human intracortical inhibitory system. Clin Neurophysiol 126(12):2314–2319. https://doi.org/10.1016/j.clinph.2015.01.030
Nojima I, Koganemaru S, Mima T (2016) Combination of static magnetic fields and peripheral nerve stimulation can alter focal cortical excitability. Front Hum Neurosci 10. https://doi.org/10.3389/fnhum.2016.00598
Dileone M, Carrasco-López MC, Segundo-Rodriguez JC, Mordillo-Mateos L, López-Ariztegui N, Alonso-Frech F et al (2017) Dopamine-dependent changes of cortical excitability induced by transcranial static magnetic field stimulation in Parkinson’s disease. Sci Rep 7(1). https://doi.org/10.1038/s41598-017-04254-y
Kufner M, Brückner S, Kammer T (2017) No modulatory effects by transcranial static magnetic field stimulation of human motor and somatosensory cortex. Brain Stimul 10(3):703–710. https://doi.org/10.1016/j.brs.2017.03.001
Dileone M, Mordillo-Mateos L, Oliviero A, Foffani G (2018) Long-lasting effects of transcranial static magnetic field stimulation on motor cortex excitability. Brain Stimul 11(4):676–688. https://doi.org/10.1016/j.brs.2018.02.005
Kirimoto H, Tamaki H, Otsuru N, Yamashiro K, Onishi H, Nojima I, Oliviero A (2018) Transcranial static magnetic field stimulation over the primary motor cortex induces plastic changes in cortical nociceptive processing. Front Hum Neurosci 12. https://doi.org/10.3389/fnhum.2018.00063
Nakagawa K, Sasaki A, Nakazawa K (2019) Accuracy in pinch force control can be altered by static magnetic field stimulation over the primary motor cortex. Neuromodulation (17). https://doi.org/10.1111/ner.12912
Sheffield A, Ahn S, Alagapan S, Fröhlich F (2018) Modulating neural oscillations by transcranial static magnetic field stimulation of the dorsolateral prefrontal cortex: a crossover, double-blind, sham-controlled pilot study. Eur J Neurosci 49(2):250–262. https://doi.org/10.1111/ejn.14232
Nojima I, Watanabe T, Gyoda T, Sugata H, Ikeda T, Mima T (2019) Transcranial static magnetic stimulation over the primary motor cortex alters sequential implicit motor learning. Neurosci Lett 696(7):33–37. https://doi.org/10.1016/j.neulet.2018.12.010
Davila-Pérez P, Pascual-Leone A, Cudeiro J (2019) Effects of transcranial static magnetic stimulation on motor cortex evaluated by different TMS waveforms and current directions. Neuroscience 413:22–30. https://doi.org/10.1016/j.neuroscience.2019.05.065
Lacroix, A., Proulx-Bégin, L., Hamel, R., De Beaumont, L., Bernier, PM., & Lepage JF. Static magnetic stimulation of the primary motor cortex impairs online but not offline motor sequence learning. Sci Rep 2019, 9(1): 1-8. https://doi.org/10.1038/s41598-019-46379-2
Kirimoto H, Tamaki H, Matsumoto T, Sugawara K, Suzuki M, Oyama M, Onishi H (2014) Effect of transcranial static magnetic field stimulation over the sensorimotor cortex on somatosensory evoked potentials in humans. Brain Stimul 7(6):836–840. https://doi.org/10.1016/j.brs.2014.09.016
Kirimoto H, Asao A, Tamaki H, Onishi H (2016) Non-invasive modulation of somatosensory evoked potentials by the application of static magnetic fields over the primary and supplementary motor cortices. Sci Rep 6(1):4–11. https://doi.org/10.1038/srep34509
Carrasco-López C, Soto-León V, Céspedes V, Profice P, Strange BA, Foffani G, Oliviero A (2017) Static magnetic field stimulation over parietal cortex enhances somatosensory detection in humans. J Neurosci 37(14):3840–3847. https://doi.org/10.1523/jneurosci.2123-16.2017
Gonzalez-Rosa JJ, Soto-Leon V, Real P, Carrasco-Lopez C, Foffani G, Strange BA, Oliviero A (2015) Static magnetic field stimulation over the visual cortex increases alpha oscillations and slows visual search in humans. J Neurosci 35(24):9182–9193. https://doi.org/10.1523/jneurosci.4232-14.2015
Rivadulla C, Aguilar J, Coletti M, Aguila J, Prieto S, Cudeiro J (2018) Static magnetic fields reduce epileptiform activity in anesthetized rat and monkey. Sci Rep 8(1). https://doi.org/10.1038/s41598-018-33808-x
Aguila J, Cudeiro J, Rivadulla C (2016) Effects of static magnetic fields on the visual cortex: reversible visual deficits and reduction of neuronal activity. Cereb Cortex 26(2):628–638. https://doi.org/10.1093/cercor/bhu228
Lozano-Soto E, Soto-León V, Sabbarese S et al (2017) Transcranial static magnetic field stimulation (tSMS) of the visual cortex decreases experimental photophobia. Cephalalgia 38(8):1493–1497. https://doi.org/10.1177/0333102417736899
Nakagawa K, Nakazawa K (2018) Static magnetic field stimulation applied over the cervical spinal cord can decrease corticospinal excitability in finger muscle. Clin Neurophysiol Pract 3:49–53. https://doi.org/10.1016/j.cnp.2018.02.001
Matsugi A (2017) Cerebellar transcranial static magnetic field stimulation transiently reduces cerebellar brain inhibition. Funct Neurol 32(2):77. https://doi.org/10.11138/fneur/2017.32.2.077
Purves D (2011) Neuroscience, 5th edn. Sunderland, Massachusetts U.S.A. Sinauer Associates Inc
Liberati A, Altman DG, Tetzlaff J et al (2009) The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: explanation and elaboration. PLoS Med 6(7):e1000100. https://doi.org/10.1016/j.jclinepi.2009.06.006
Moher D, Liberati A, Tetzlaff J, Altman DG (2009) Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. Ann Intern Med 151(4):264e9. https://doi.org/10.1136/bmj.b2535
Yamato TP, Maher C, Koes B, Moseley A (2017) The PEDro scale had acceptably high convergent validity, construct validity, and interrater reliability in evaluating methodological quality of pharmaceutical trials. J Clin Epidemiol 86:176–181. https://doi.org/10.1016/j.jclinepi.2017.03.002
Hooijmans, C. R., Rovers, M. M., de Vries, R. B., Leenaars, M., Ritskes-Hoitinga, M., & Langendam, M. W. SYRCLE’s risk of bias tool for animal studies. BMC Med Res Methodol 2014, 14(1):43. https://doi.org/10.1186/1471-2288-14-43
Banerjee J, Sorrell ME, Celnik PA, Pelled G (2017) Immediate effects of repetitive magnetic stimulation on single cortical pyramidal neurons. PLoS One 12(1). https://doi.org/10.1371/journal.pone.0170528
Huerta PT, Volpe BT (2009) Transcranial magnetic stimulation, synaptic plasticity and network oscillations. J NeuroEng Rehabil 6:7. https://doi.org/10.1186/1743-0003-6-7
Bikson M, Inoue M, Akiyama H et al (2004) Effects of uniform extracellular DC electric fields on excitability in rat hippocampal slices in vitro. J Physiol 557(Pt 1):175–190. https://doi.org/10.1113/jphysiol.2003.055772
Zaghi S, Acar M, Hultgren B, Boggio PS, Fregni F (2010) Noninvasive brain stimulation with low-intensity electrical currents: putative mechanisms of action for direct and alternating current stimulation. Neuroscientist. 16(3):285–307. https://doi.org/10.1177/1073858409336227
Kim S, Chung Y-A, Lee C-U, Chae J-H, Juh R, Jeong J (2010) Target-specific rCBF changes induced by 0.3-T static magnetic field exposure on the brain. Brain Res 1317:211–217. https://doi.org/10.1016/j.brainres.2009.10.057
Oliviero A, Carrasco-López MC, Campolo M et al (2015) Safety study of transcranial static magnetic field stimulation (tSMS) of the human cortex. Brain Stimul 8(3):481–485. https://doi.org/10.1016/j.brs.2014.12.002
Bertolino G, Dutra Souza HC, de Araujo JE (2013) Neuropathology and behavioral impairments in Wistar rats with a 6-OHDA lesion in the substantia nigra compacta and exposure to a static magnetic field. Electromagn Biol Med 32(4):527–535. https://doi.org/10.3109/15368378.2012.751394
McLean MJ, Engström S, Holcomb RR, Sanchez D (2003) A static magnetic field modulates severity of audiogenic seizures and anticonvulsant effects of phenytoin in DBA/2 mice. Epilepsy Res 55(1-2):105–116. https://doi.org/10.1016/s0920-1211(03)00109-8
Giorgetto, C., Silva, E. C. M., Kitabatake, T. T., Bertolino, G., & de Araujo, J. E. Behavioural profile of Wistar rats with unilateral striatal lesion by quinolinic acid (animal model of Huntington disease) post-injection of apomorphine and exposure to static magnetic field. Exp Brain Res 2015, 233(5): 1455–1462. https://doi.org/10.1007/s00221-015-4219-7
Di Lazzaro V, Rothwell JC, Oliviero A et al (1999) Intracortical origin of the short latency facilitation produced by pairs of threshold magnetic stimuli applied to human motor cortex. Exp Brain Res 129:494–499. https://doi.org/10.1007/s002210050919
Di Lazzaro V, Restuccia D, Oliviero A et al (1998) Magnetic transcranial stimulation at intensities below active motor threshold activates intracortical inhibitory circuits. Exp Brain Res 119:265–268. https://doi.org/10.1007/s002210050341
Ugawa Y, Uesaka Y, Terao Y, Hanajima R, Kanazawa I (1995) Magnetic stimulation over the cerebellum in humans. Ann Neurol 37(6):703–713. https://doi.org/10.1002/ana.410370603
Rivadulla C, Foffani G, Oliviero A (2013) Magnetic field strength and reproducibility of neodymium magnets useful for transcranial static magnetic field stimulation of the human cortex. Neuromodulation 17(5):438–442. https://doi.org/10.1111/ner.12125
Tharayil JJ, Goetz SM, Bernabei JM, Peterchev AV (2017) Field distribution of transcranial static magnetic stimulation in realistic human head model. Neuromodulation 21(4):340–347. https://doi.org/10.1111/ner.12699
Nakano M, Yamada S, Udagawa R, Kato N (2004) Frequency dependent requirement for calcium store-operated mechanisms in induction of homosynaptic long-term depression at hippocampus CA1 synapses. Eur J Neurosci 19:2881–2887. https://doi.org/10.1111/j.0953-816X.2004.03390.x
Pinto AD, Chen R (2001) Suppression of the motor cortex by magnetic stimulation of the cerebellum. Exp Brain Res 140(4):505–510. https://doi.org/10.1007/s002210100862
Daskalakis ZJ, Paradiso GO, Christensen BK, Fitzgerald PB, Gunraj C, Chen R (2004) Exploring the connectivity between the cerebellum and motor cortex in humans. J Physiol 557(2):689–700. https://doi.org/10.1113/jphysiol.2003.059808
Iwata N, Ugawa Y (2005) The effects of cerebellar stimulation on the motor cortical excitability in neurological disorders: a review. Cerebellum 4(4):218–223. https://doi.org/10.1080/14734220500277007
Ben Yakir-Blumkin M, Loboda Y, Schächter L, Finberg JPM (2014) Neuroprotective effect of weak static magnetic fields in primary neuronal cultures. Neuroscience 278:313–326. https://doi.org/10.1016/j.neuroscience.2014.08.029
Abraham WC, Bear MF (1996) Metaplasticity: the plasticity of synaptic plasticity. Trends Neurosci 19(4):126–130. https://doi.org/10.1016/S0166-2236(96)80018-X
Isaacson JS, Scanziani M (2011) How inhibition shapes cortical activity. Neuron. 72(2):231–243. https://doi.org/10.1016/j.neuron.2011.09.027
Chen SX, Kim AN, Peters AJ, Komiyama T (2015) Subtype-specific plasticity of inhibitory circuits in motor cortex during motor learning. Nat Neurosci 18(8):1109–1115. https://doi.org/10.1038/nn.4049
Kuhn YA, Keller M, Ruffieux J, Taube W (2017) Intracortical inhibition within the primary motor cortex can be modulated by changing the focus of attention. J Vis Exp (127):55771. https://doi.org/10.3791/55771
Hummel FC, Steven B, Hoppe J et al (2009) Deficient intracortical inhibition (SICI) during movement preparation after chronic stroke. Neurology 72(20):1766–1772. https://doi.org/10.1212/WNL.0b013e3181a609c5
Mall V, Berweck S, Fietzek UM et al (2004) Low level of intracortical inhibition in children shown by transcranial magnetic stimulation. Neuropediatrics 35(2):120–125. https://doi.org/10.1055/s-2004-815834
Papegaaij S, Taube W, Baudry S, Otten E, Hortobágyi T (2014) Aging causes a reorganization of cortical and spinal control of posture. Front Aging Neurosci 6:28. https://doi.org/10.3389/fnagi.2014.00028
Xue M, Atallah BV, Scanziani M (2014) Equalizing excitation-inhibition ratios across visual cortical neurons. Nature. 511(7511):596–600. https://doi.org/10.1038/nature13321
Kolasinski J, Hinson EL, Divanbeighi Zand AP, Rizov A, Emir UE, Stagg CJ (2018) The dynamics of cortical GABA in human motor learning. J Physiol. https://doi.org/10.1113/JP276626
Floyer-Lea A, Wylezinska M, Kincses T, Matthews PM (2006) Rapid modulation of GABA concentration in human sensorimotor cortex during motor learning. J Neurophysiol 95(3):1639–1644. https://doi.org/10.1152/jn.00346.2005
Thut G (2006) Band electroencephalographic activity over occipital cortex indexes visuospatial attention bias and predicts visual target detection. J Neurosci 26(37):9494–9502. https://doi.org/10.1523/jneurosci.0875-06.2006
Donner TH, Kettermann A, Diesch E, Ostendorf F, Villringer A, Brandt SA (2002) Visual feature and conjunction searches of equal difficulty engage only partially overlapping frontoparietal networks. NeuroImage 15(1):16–25. https://doi.org/10.1006/nimg.2001.0951
Min B-K, Herrmann CS (2007) Prestimulus EEG alpha activity reflects prestimulus top-down processing. Neurosci Lett 422(2):131–135. https://doi.org/10.1016/j.neulet.2007.06.013
Kawasaki M, Kitajo K, Yamaguchi Y (2014) Fronto-parietal and fronto-temporal theta phase synchronization for visual and auditory-verbal working memory. Front Psychol 5:1–7. https://doi.org/10.3389/fpsyg.2014.00200
Scholz S, Schneider SL, Rose M (2017) Differential effects of ongoing EEG beta and theta power on memory formation. PLoS ONE 12(2):1–18. https://doi.org/10.1371/journal.pone.0171913
Khanna P, Carmena JM (2017) Beta band oscillations in motor cortex reflect neural population signals that delay movement onset. eLife 6. https://doi.org/10.7554/eLife.24573
Hofman D, Schutter DJLG (2011) Asymmetrical frontal resting-state beta oscillations predict trait aggressive tendencies and behavioral inhibition. Soc Cogn Affect Neurosci 7(7):850–857. https://doi.org/10.1093/scan/nsr060
Ferrara JM, Adam OR, Ondo WG (2010) Levodopa-induced dyskinesias in spinocerebellar ataxia type 2. Arch Neurol 67(1):114–115. https://doi.org/10.1001/archneurol.2009.291
Wang X, Mao Z, Yu X (2020) The role of noninvasive brain stimulation for behavioral and psychological symptoms of dementia: a systematic review and meta-analysis. Neurol Sci 2020(41):1063–1074. https://doi.org/10.1007/s10072-020-04245-4
Alomar AS, Saeedi JR (2020) Different modalities of invasive neurostimulation for epilepsy. Neurol Sci. https://doi.org/10.1007/s10072-020-04614-z
Wang Z, Chen J, Lin Z et al (2020) Transcranial direct current stimulation improves the swallowing function in patients with cricopharyngeal muscle dysfunction following a brainstem stroke. Neurol Sci 41:569–574. https://doi.org/10.1007/s10072-019-04120-x
Trebossen V, Bouaziz N, Benadhira R, Januel D (2017) Transcranial direct current stimulation for patients with benign essential blepharospasm: a case report. Neurol Sci 38(1):201–202. https://doi.org/10.1007/s10072-016-2703-x
Acknowledgements
EV and MD want to express their gratitude to Drs. Juana Gallar and María del Carmen Acosta for kindly allowing us time to contribute to this review while working on other projects. Also, we wanted to acknowledge Dr. Santiago Canals for his willingness to share scientific knowledge and interesting discussions. Finally, we want to acknowledge Dr. Antonio Oliviero for letting NV visit his lab and learn from the latest trends and usage of tSMS from its very first source.
Availability of data and material
All the data reviewed in this study have been extracted from the articles included in the tables. The search strategy is indicated both in the “Methods” section of this document and in Fig. 2. Furthermore, this figure includes the reasons for exclusion of some articles. The list of excluded studies based on full text analysis is available on request by investigators.
Code availability
Due to the early stage of tSMS intervention and the available data on its effects, a systematic review registration number is not available.
Author information
Authors and Affiliations
Contributions
Nuria Viudes-Sarrion participated in design of study, data acquisition, data analysis and interpretation, and drafting of the manuscript; Enrique Velasco participated in design of study, data acquisition, data analysis and interpretation, and drafting of the manuscript; Miguel Delicado-Miralles participated in data acquisition, data analysis, and drafting of the manuscript; Carmen Lillo-Navarro participated in design and conceptualization of study, data interpretation, and drafting and revising of the manuscript.
Corresponding author
Ethics declarations
Ethical approval
The article is based upon previously published studies and patient data and is in line with the journal’s ethical guidelines.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
ESM 1
(PDF 797 kb)
Glossary
- TMS
-
Transcranial magnetic stimulation
- tDCS
-
Transcranial direct current stimulation
- EEG
-
Electroencephalography
- MEP
-
Motor-evoked potential
- SICI
-
Short-interval intracortical inhibition
- SICF
-
Short-interval intracortical facilitation
- LTD
-
Long-term depression
- LTP
-
Long-term potentiation
- RMTs
-
Resting motor thresholds
- DLPFC
-
Dorsolateral prefrontal cortex
- PNS
-
Peripheral nerve stimulation
- PV
-
Parvalbumin
- SEP
-
Somatosensory-evoked potential
- FDI
-
First digital interosseous muscle
- RT
-
Reaction time
- RR
-
Rotarod test
- Cylindrical NdFeB magnet
-
Cylindrical nickel-plated (Ni–Cu–Ni) NdFeB magnet
- APB
-
Abductor pollicis brevis muscle
- ADM
-
Abductor digiti minimi muscle
- Online learning
-
Learning during the motor task
- Offline learning
-
Learning after the completion of the motor task between sessions
- Implicit motor learning
-
Improvement in sequence performance without knowing the sequence
- Explicit motor learning
-
Improvement in sequence performance knowing the sequence
Rights and permissions
About this article
Cite this article
Viudes-Sarrion, N., Velasco, E., Delicado-Miralles, M. et al. Static magnetic stimulation in the central nervous system: a systematic review. Neurol Sci 42, 1733–1749 (2021). https://doi.org/10.1007/s10072-021-05156-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10072-021-05156-8