Abstract
Purposes
Attempts have been made to use bevacizumab (BEV) in an adjuvant or neoadjuvant setting. However, BEV is known to cause various adverse events, and the safety of neoadjuvant BEV has not yet been fully evaluated. This study assessed the postoperative complications in patients receiving neoadjuvant BEV for colorectal cancer.
Methods
The data for 78 patients with resectable advanced or metastatic colorectal cancer who received neoadjuvant BEV followed by surgical resection were retrospectively analyzed.
Results
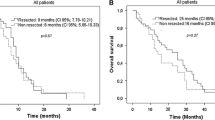
The median interval between the last BEV dose and surgery was 9 weeks. The most common postoperative complication was pelvic sepsis, which occurred in 11 patients (14 %). A biliary fistula developed in four of 23 patients who underwent liver resection. Anastomotic leakage occurred in six of 24 patients with a colorectal anastomosis, four of whom required re-laparotomy. In a univariate analysis, male gender and a greater intraoperative blood loss were associated with postoperative complications of any grade. Colorectal anastomosis was a risk factor for major complications. In a multivariate analysis, intraoperative blood loss was an independent risk factor for postoperative complications of any grade (HR 6.338; P = 0.003). With regard to major postoperative complications, colorectal primary anastomosis was the only independent predictive risk factor (HR 8.285; P = 0.013).
Conclusions
In patients with colorectal cancer who underwent elective surgery after BEV treatment, the interval between BEV and surgery was not a risk factor for postoperative complications (based on a median interval of 9 weeks). Colorectal primary anastomosis was the only independent risk factor for major postoperative complications.
Similar content being viewed by others
References
Foundation for promotion of cancer research. The editorial board of the cancer statistics in Japan. Cancer statistics in Japan–2009. Tokyo, Japan.
Kopetz S, Chang GJ, Overman MJ, Eng C, Sargent DJ, Larson DW, et al. Improved survival in metastatic colorectal cancer is associated with adoption of hepatic resection and improved chemotherapy. J Clin Oncol. 2009;27:3677–83.
Hurwitz H, Fehrenbacher L, Novotny W, Cartwright T, Hainsworth J, Heim W, et al. Bevacizumab plus irinotecan, fluorouracil, and leucovorin for metastatic colorectal cancer. N Engl J Med. 2004;350:2335–42.
Kabbinavar FF, Flynn PJ, Kozloff M, Ashby MA, Sing A, Barr CE, et al. Gastrointestinal perforation associated with bevacizumab use in metastatic colorectal cancer: results from a large treatment observational cohort study. Eur J Cancer. 2012;48:1126–32.
Kozloff M, Yood MU, Berlin J, Flynn PJ, Kabbinavar FF, Purdie DM, et al. Clinical outcomes associated with bevacizumab-containing treatment of metastatic colorectal cancer: the BRiTE observational cohort study. Oncologist. 2009;14:862–70.
Van Cutsem E, Rivera F, Berry S, Kretzschmar A, Michael M, DiBartolomeo M, et al. Safety and efficacy of first-line bevacizumab with FOLFOX, XELOX, FOLFIRI and fluoropyrimidines in metastatic colorectal cancer: the BEAT study. Ann Oncol. 2009;20:1842–7.
Hapani S, Chu D, Wu S. Risk of gastrointestinal perforation in patients with cancer treated with bevacizumab: a meta-analysis. Lancet Oncol. 2009;10:559–68.
Saito S, Hayashi N, Sato N, Iwatsuki M, Baba Y, Sakamoto Y, et al. Chemotherapy with bevacizumab for metastatic colorectal cancer; a retrospective review of 181 Japanese patients. Int J Clin Oncol. 2012; (Epub ahead of print).
Scappaticci FA, Fehrenbacher L, Cartwright T, Hainsworth JD, Heim W, Berlin J, et al. Surgical wound healing complications in metastatic colorectal cancer patients treated with bevacizumab. J Surg Oncol. 2005;91:173–80.
August DA, Serrano D, Poplin E. “Spontaneous,” delayed colon and rectal anastomotic complications associated with bevacizumab therapy. J Surg Oncol. 2008;97:180–5.
Bège T, Lelong B, Viret F, Turrini O, Guiramand J, Topart D, et al. Bevacizumab-related surgical site complication despite primary tumor resection in colorectal cancer patients. Ann Surg Oncol. 2009;16:856–60.
Lordan JT, Wilkins M, Karanjia ND. Delayed bile leak with avastin after liver resection for metastatic colorectal cancer. Hepatogastroenterology. 2011;58:1769–70.
Itatani Y, Akiyoshi T, Kuroyanagi H, Yamakawa K, et al. Total mesorectal excision of initially unresectable locally advanced rectal cancer infiltrating the pelvic wall after treatment with FOLFOX4 plus bevacizumab and preoperative chemoradiation: report of a case. Surg Today. 2012;42:75–9.
Kesmodel SB, Ellis LM, Lin E, Chang GJ, Abdalla EK, Kopetz S, et al. Preoperative bevacizumab does not significantly increase postoperative complication rates in patients undergoing hepatic surgery for colorectal cancer liver metastases. J Clin Oncol. 2008;26:5254–60.
Reddy SK, Morse MA, Hurwitz HI, Bendell JC, Gan TJ, Hill SE, et al. Addition of bevacizumab to irinotecan- and oxaliplatin-based preoperative chemotherapy regimens does not increase morbidity after resection of colorectal liver metastases. J Am Coll Surg. 2008;206:96–106.
Tamandl D, Gruenberger B, Klinger M, Herberger B, Kaczirek K, Fleischmann E, et al. Liver resection remains a safe procedure after neoadjuvant chemotherapy including bevacizumab: a case-controlled study. Ann Surg. 2010;252:124–30.
Mahfud M, Breitenstein S, El-Badry AM, Puhan M, Rickenbacher A, Samaras P, et al. Impact of preoperative bevacizumab on complications after resection of colorectal liver metastases: case-matched control study. World J Surg. 2010;34:92–100.
National comprehensive cancer network. NCCN clinical practice guidelines in oncology. Colon cancer v.1. 2013. http://www.nccn.org/professionals/physician_gls/pdf/colon.pdf. Accessed 15 Sep 2012.
Schrag D, Weiser MR, Goodman KA, Gonen M, Cercek A, Reidy DL, et al. Neoadjuvant FOLFOX-bev, without radiation, for locally advanced rectal cancer. J Clin Oncol. 2010; 28:15s suppl (abstr 3511).
Fernandez-Martos C, Estevan R, Salud A, Pericay C, Gallen M, Sierra E, et al. Neoadjuvant capecitabine, oxliplatin, and bevacizumab (CAPOX-B) in intermediate-risk rectal cancer (RC) patients defined by magnetic resonance (MR): GEMCAD 0801 trial. J Clin Oncol. 2012; 30 suppl (abstr 3586).
Uehara K, Ishiguro S, Sakamoto E, Maeda A, Inoue M, Tojima Y, et al. Phase II trial of neoadjuvant chemotherapy with XELOX plus bevacizumab for locally advanced rectal cancer. Jpn J Clin Oncol. 2011;41:1041–4.
Crane CH, Eng C, Feig BW, Das P, Skibber JM, Chang GJ, et al. Phase II trial of neoadjuvant bevacizumab, capecitabine, and radiotherapy for locally advanced rectal cancer. Int J Radiat Oncol Biol Phys. 2010;76:824–30.
Dindo D, Demartines N, Clavien PA. Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg. 2004;240:205–13.
Japanese society for cancer of the colon and rectum. Japanese classification of colorectal carcinoma. Second english edition.
Ribero D, Wang H, Donadon M, Zorzi D, Thomas MB, Eng C, et al. Bevacizumab improves pathologic response and protects against hepatic injury in patients treated with oxaliplatin-based chemotherapy for colorectal liver metastases. Cancer. 2007;110:2761–7.
Klinger M, Eipeldauer S, Hacker S, Herberger B, Tamandl D, Dorfmeister M, et al. Bevacizumab protects against sinusoidal obstruction syndrome and does not increase response rate in neoadjuvant XELOX/FOLFOX therapy of colorectal cancer liver metastases. Eur J Surg Oncol. 2009;35:515–20.
van der Pool AE, Marsman HA, Verheij J, Ten Kate FJ, Eggermont AM, Ijzermans JN, et al. Effect of bevacizumab added preoperatively to oxaliplatin on liver injury and complications after resection of colorectal liver metastases. J Surg Oncol. 2012;106:892–7.
Nordlinger B, Sorbye H, Glimelius B, Poston GJ, Schlag PM, Rougier P, et al. Perioperative chemotherapy with FOLFOX4 and surgery versus surgery alone for resectable liver metastases from colorectal cancer (EORTC Intergroup trial 40983): a randomised controlled trial. Lancet. 2008;371:1007–16.
Starlinger P, Alidzanovic L, Schauer D, Maier T, Nemeth C, Perisanidis B, et al. Neoadjuvant bevacizumab persistently inactivates VEGF at the time of surgery despite preoperative cessation. Br J Cancer. 2012;107:961–6.
Conflict of interest
Yuichiro Yoshioka and co-authors have no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yoshioka, Y., Uehara, K., Ebata, T. et al. Postoperative complications following neoadjuvant bevacizumab treatment for advanced colorectal cancer. Surg Today 44, 1300–1306 (2014). https://doi.org/10.1007/s00595-013-0686-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00595-013-0686-2