Abstract
Aim
The activation of Hedgehog signaling, which is critical to normal mammalian gastrointestinal development, is implicated in the development of various tumors, including colorectal cancer. In the pancreas, a precursor lesion overexpresses the Sonic hedgehog (Shh) when compared with normal tissue and cancer. The present study was designed to investigate Shh related protein expression in hyperplastic polyps and the adenoma-carcinoma sequence in the colon and rectum.
Methods
Seventeen hyperplastic polyps, 24 adenomas of the colon, 69 adenocarcinomas (31 well-differentiated, 38 moderately-differentiated), and 30 normal colon samples were used in the study. We checked the expression of Shh, both patched (Ptch) and smoothened (Smo), by immunohistochemistry and compared the expression rate of each group.
Results
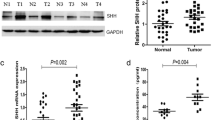
Almost all adenomas, 22 of 23 (96%), expressed Shh. In other groups, 4 of 17 hyperplastic polyps (24%), 7 of 31 well-differentiated adenocarcinomas (23%), 13 of 38 moderately-differentiated adenocarcinomas (34%) and none of the 30 normal samples expressed Shh. The rate of expression in Ptch and Smo gradually increased in accordance with tumor progression.
Conclusion
This result indicates that Shh-related carcinogenesis and Shh expression may be a trigger for the adenoma-carcinoma sequence. This study suggests a potential therapeutic target of hedgehog blockade in carcinogenesis.



Similar content being viewed by others

Abbreviations
- Shh:
-
Sonic hedgehog
- Ptch:
-
Patched
- Smo:
-
Smoothened
References
Lees C, Howie S, Sartor RB, Satsangi J. The hedgehog signalling pathway in the gastrointestinal tract: implications for development, homeostasis, and disease. Gastroenterology. 2005;129:1696–710.
Fukaya M, Isohata N, Ohta H, Aoyagi K, Ochiya T, Saeki N, et al. Hedgehog signal activation in gastric pit cell and in diffuse-type gastric cancer. Gastroenterology. 2006;131:14–29.
Douard R, Moutereau S, Pernet P, Chimingqi M, Allory Y, Manivet P, et al. Sonic hedgehog-dependent proliferation in a series of patients with colorectal cancer. Surgery. 2006;139:665–70.
Ma X, Chen K, Huang S, Zhang X, Adegboyega PA, Evers BM, et al. Frequent activation of the hedgehog pathway in advanced gastric adenocarcinomas. Carcinogenesis. 2005;26:1698–705.
Thayar SP, Magliano MP, Heiser PW, Nielsen CM, Roberts DJ, Lauwers GY, et al. Hedgehog is an early and late mediator of pancreatic cancer tumorigenesis. Nature. 2003;23:851–6.
Ohuchida K, Mizumoto K, Fujita H, Yamaguchi H, Konomi H, Nagai E, et al. Sonic hedgehog is an early developmental marker of intraductal papillary mucinous neoplasms: clinical implications of mRNA levels in pancreatic juice. J Pathol. 2006;210:42–8.
Prased NB, Biankin AV, Fukushima N, Maitra A, Dhara S, Elkahloun AG, et al. Gene expression profiles in pancreatic intraepithelial neoplasia reflect the effects of hedgehog signaling on pancreatic ductal epithelial cells. Cancer Res. 2005;65:1619–25.
Magliano MP, Sekine S, Ermilov A, Ferris J. Hedgehog/Ras interactions regulate early stages of pancreatic cancer. Gene Dev. 2006;20:3161–73.
Beachy PA, Karhadkar SS, Berman DM. Tissue repair and stem cell renewal in carcinogenesis. Nature. 2004;432:324–31.
Evangelista M, Tian H, Sauvage FJ. The hedgehog signaling pathway in cancer. Clin Cancer Res. 2006;12:5924–8.
Nakashima H, Nakamura M, Yamaguchi H, Yamanaka N, Akiyoshi T, Koga K, et al. Nuclear Factor-κB contributes to hedgehog signaling pathway activation through sonic hedgehog induction in pancreatic cancer. Cancer Res. 2006;66:7041–9.
Cutclife C, Kersey D, Huang C-C, Zeng Y, Walterhouse D, Perlman EJ. Clear cell sarcoma of the kidney: up-regulation of neural markers with activation of the sonic hedgehog and Akt pathways. Clin Cancer Res. 2005;11:7986–93.
Ma X, Sheng T, Zhang Y, He J, Huang S, et al. Hedgehog signaling is activated in subsets of esophageal cancers. Int J Cancer. 2006;118:139–48.
Luebeck EG, Moolgavkar SH. Multistage carcinogenesis and the incidence of colorectal cancer. PNAS. 2002;12:15095–100.
Ohta M, Tateishi K, Kanai F, Watabe H, Kondo S, Guleng B, et al. p53-Independent negative regulation of p21/cyclin dependent kinase interacting protein 1 by the sonic hedgehog glioma associated oncogene 1 pathway in gastric carcinoma cells. Cancer Res. 2005;65:10822–9.
Oniscu A, James RM, Morris RG, Bader S, Malcomson RD, Harrison D. Expression of Sonic hedgehog pathway genes is altered in colonic neoplasia. J Pathol. 2004;203:909–17.
Sims-Mourtada J, Izzo JG, Apisarnthanarax S, Wu T-T, Malhotra U, Luthra R, et al. Hedgehog: an attribute to tumor regrowth after chemoradiotherapy and a target to improve radiation response. Clin Cancer Res. 2006;12:6565–72.
Qualtrough D, Buda A, Gaffield W, Williams AC, Paraskeva C. Hedgehog signaling in colorectal tumour cells: induction of apoptosis with cyclopamine treatment. Int J Cancer. 2004;110:831–7.
Ferrandez A, Samowitz W, DiSario JA, Burt RW. Phenotypic characteristics and risk of cancer development in hyperplastic polyposis: case series and literature review. Am J Gastroenterol. 2004;99:2012–8.
Wynter CVA, Walsh MD, Higuchi T, Leggett BA, Young J, Jass JR. Methylation patterns define two types of hyperplastic polyp associated with colorectal cancer. GUT. 2004;53:573–80.
Jass JR. Hyperplastic polyps of the colorectum-innocent or guilty? Dis Colon Rectum. 2001;44:163–6.
Zauber P, Sabbath-Solitare M, Marotta S, Zauber A., Bishop T. Comparative molecular pathology of sporadic hyperplastic polyps and neoplastic lesions from the same individual. J Clin Pathol. 2004;57:1084–8.
Acknowledgments
This study was supported by the grant of Cancer Research Project Cooperated by TAIHO Pharmaceutical Co., Ltd. and The University of Tokushima.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yoshikawa, K., Shimada, M., Miyamoto, H. et al. Sonic hedgehog relates to colorectal carcinogenesis. J Gastroenterol 44, 1113–1117 (2009). https://doi.org/10.1007/s00535-009-0110-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00535-009-0110-2