Abstract
Background and aims
Neoadjuvant chemotherapy is increasingly being used to enlarge the cohort of patients who can be offered hepatic resection for malignancy. However, the impact of these agents on the liver parenchyma itself, and their effects on clinical outcomes following hepatic resection remain unclear. This review identifies patterns of regimen-specific chemotherapy-induced hepatic injury and assesses their impact on outcomes following hepatic resection for colorectal liver metastases (CLM).
Methods
An electronic search was performed using the MEDLINE (US Library of Congress) database from 1966 to May 2007 to identify relevant articles related to chemotherapy-induced hepatic injury and subsequent outcome following hepatic resection.
Results
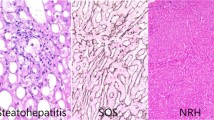
The use of the combination of 5-flourouracil and leucovorin is linked to the development of hepatic steatosis, and translates into increased postoperative infection rates. A form of non-alcoholic steatohepatitis (NASH) related to chemotherapy and otherwise known as chemotherapy-associated steatohepatitis (CASH) is closely linked to irinotecan-based therapy and is associated with inferior outcomes following hepatic surgery mainly due to hepatic insufficiency and poor regeneration. Data on sinusoidal obstruction syndrome (SOS) following treatment with oxaliplatin are less convincing, but there appears to be an increased risk for intra-operative bleeding and decreased hepatic reserve associated with the presence of SOS. Intra-arterial floxuridine therapy damages the extrahepatic biliary tree in addition to causing parenchymal liver damage, and has been shown to be associated with increased morbidity after hepatic resection.
Conclusion
Agent-specific patterns of damage are now being recognized with increasing use of neoadjuvant chemotherapy prior to surgery. The potential benefits and risks of these should be considered on an individual patient basis prior to hepatic resection.
Similar content being viewed by others
References
Pawlik TM, Scoggins CR, Zorzi D, et al. Effect of surgical margin status on survival and site of recurrence after hepatic resection for colorectal metastases. Ann Surg. 2005;241:715–22.
Fernandez FG, Drebin JA, Linehan DC, Dehdashti F, Siegel BA, Strasberg SM. Five-year survival after resection of hepatic metastases from colorectal cancer in patients screened by positron emission tomography with F-18 fluorodeoxyglucose (FDG-PET). Ann Surg. 2004;240:438–47.
Choti MA, Sitzmann JV, Tiburi MF, et al. Trends in long-term survival following liver resection for hepatic colorectal metastases. Ann Surg. 2002;235:759–66.
Abdalla EK, Vauthey JN, Ellis LM, et al. Recurrence and outcomes following hepatic resection, radiofrequency ablation, and combined resection/ablation for colorectal liver metastases. Ann Surg. 2004;239:818–25.
Lee WS, Yun HR, Yun SH, Chun HK, Lee WY, Kim SJ, Choi SH, Heo JS, Joh JW, Park YS, Kang WK. Treatment outcomes of hepatic and pulmonary metastases from colorectal carcinoma. J Gastroenterol Hepatol. 2007;PMID: 18086122.
Arru M, Aldrighetti L, Castoldi R, et al. Analysis of prognostic factors influencing long-term survival after hepatic resection for metastatic colorectal cancer. World J Surg. 2008;32(1):93–103.
Taniai N, Akimaru K, Yoshida H, Tajiri T. Surgical treatment for better prognosis of patients with liver metastases from colorectal cancer. Hepatogastroenterology. 2007;54(78):1805–9.
Lau WY, Lai EC. Hepatic resection for colorectal liver metastases. Singapore Med J. 2007;48(7):635–9.
Saltz LB, Cox JV, Blanke C, et al. Irinotecan plus fluorouracil and leucovorin for metastatic colorectal cancer. Irinotecan study group. N Engl J Med. 2000;343:905–14.
Tournigand C, Andre T, Achille E, et al. FOLFIRI followed by FOLFOX6 or the reverse sequence in advanced colorectal cancer: a randomized GERCOR study. J Clin Oncol. 2004;22:229–37.
Douillard JY, Cunningham D, Roth AD, et al. Irinotecan combined with fluorouracil compared with fluorouracil alone as first-line treatment for metastatic colorectal cancer: a multicentre randomised trial. Lancet. 2000;355:1041–7.
de Gramont A, Figer A, Seymour M, et al. Leucovorin and fluorouracil with or without oxaliplatin as first- line treatment in advanced colorectal cancer. J Clin Oncol. 2000;18:2938–47.
Levi F, Zidani R, Misset JL. Randomised multicentre trial of chronotherapy with oxaliplatin, fluorouracil, and folinic acid in metastatic colorectal cancer. International Organization for Cancer Chronotherapy. Lancet. 1997;350:681–6.
Hoff PM, Eng C, Adinin RB, et al. Preliminary results from a phase II study of FOLFIRI plus bevacizumab as first-line treatment for metastatic colorectal cancer (mCRC). ASCO annual meeting proceedings 2006;24(Suppl):165S.
Diaz Rubio E, Tabernero J, Van Cutsem E, et al. Cetuximab in combination with oxaliplatin/5-fluorouracil (5-FU)/folinic acid (FA) (FOLFOX-4) in the first-line treatment of patients with epidermal growth factor receptor (EGFR)-expressing metastatic colorectal cancer: an international phase II study. ASCO annual meeting proceedings. J Clin Oncol 2005;23(Suppl):254s.
Jaeck D, Oussoultzoglou E, Rosso E, Greget M, Weber JC, Bachellier P. A two-stage hepatectomy procedure combined with portal vein embolization to achieve curative resection for initially unresectable multiple and bilobar colorectal liver metastases. Ann Surg. 2004;240:1037–49.
Abdalla EK, Barnett CC, Doherty D, Curley SA, Vauthey JN. Extended hepatectomy in patients with hepatobiliary malignancies with and without preoperative portal vein embolization. Arch Surg. 2002;137:675–80.
Adam R, Pascal G, Castaing D, et al. Tumor progression while on chemotherapy: a contraindication to liver resection for multiple colorectal metastases? Ann Surg. 2004;240:1052–61.
Adam R, Delvart V, Pascal G, et al. Rescue surgery for unresectable colorectal liver metastases downstaged by chemotherapy: a model to predict long-term survival. Ann Surg. 2004;240:644–57.
Nordlinger B, Sorbye H, Collette L, et al. Final results of the EORTC intergroup randomized phase III study 40983 [EPOC] evaluating the benefit of perioperative FOLFOX4 chemotherapy for patients with potentially resectable colorectal cancer liver metastases. J Clin Oncol. 2007;25:LBA5.
Bismuth H, Adam R, Lévi F, et al. Resection of nonresectable liver metastases from colorectal cancer after neoadjuvant chemotherapy. Ann Surg. 1996;224:509–20.
Gruenberger T, Sorbye H, Dubois M, et al. Tumour response to preoperative chemotherapy (CT) with FOLFOX-4 for resectable colorectal cancer liver metastases (LM). Interim results of EORTC intergroup randomised phase III trial. J Clin Oncol. 2006;24:3500.
ESMO Guidelines Working Group. Advanced colorectal cancer: ESMO clinical recommendations for diagnosis, treatment and follow-up. Ann Oncol. 2007;18(Suppl 2):25–6.
National Institute for Health and Clinic Excellence. Irinotecan, oxaliplatin and raltitrexed for the treatment of advanced colorectal cancer. http://www.nice.org.uk/TA093guidance.
Parikh AA, Gentner B, Wu TT, Curley SA, Ellis LM, Vauthey JN. Perioperative complications in patients undergoing major liver resection with or without neoadjuvant chemotherapy. J Gastrointest Surg. 2003;7:1082–88.
Lygidakis NJ, Sgourakis G, Vlachos L, Raptis S, Safioleas M, Boura P, et al. Metastatic liver disease of colorectal origin: the value of locoregional immunochemotherapy combined with systemic chemotherapy following liver resection. Results of a prospective randomized study. Hepatogastroenterology. 2001;48:1685–91.
Gallagher DJ, Capanu M, Raggio G, Kemeny N. Hepatic arterial infusion plus systemic irinotecan in patients with unresectable hepatic metastases from colorectal cancer previously treated with systemic oxaliplatin: a retrospective analysis. Ann Oncol. 2007;12:1995–9.
Sherlock S. Acute fatty liver of pregnancy and the microvesicular fat diseases. Gut. 1983;24:260–9.
D’Alessandro A, Kalayoglu M, Sollinger H, et al. The predictive value of donor liver biopsies for the development of primary nonfunction after orthotopic liver transplantation. Transplantation. 1991;51:157–63.
Ludwig J, Viggiano TR, McGIll DB, et al. Nonalcoholic steatohepatitis: mayo clinic experiences with a hitherto unnamed disease. Mayo Clin Proc. 1980;55:434–8.
Angulo P. Nonalcoholic fatty liver disease. N Engl J Med. 2002;346:1221–31.
Kooby DA, Fong Y, Suriawinata A, et al. Impact of steatosis on perioperative outcome following hepatic resection. J Gastrointest Surg. 2003;7:1034–44.
Laurent A, Nicco C, Chereau C, et al. Controlling tumor growth by modulating endogenous production of reactive oxygen species. Cancer Res. 2005;65:948–56.
Behrns KE, Tsiotos GG, DeSouza NF, Krishna MK, Ludwig J, Nagorney DM. Hepatic steatosis as a potential risk factor for major hepatic resection. J Gastrointest Surg. 1998;2:292–8.
Begriche K, Igoudjil A, Pessayre D, Fromenty B. Mitochondrial dysfunction in NASH: causes, consequences and possible means to prevent it. Mitochondrion. 2006;6:1–28.
Belghiti J, Hiramatsu K, Benosit S, Massault PP, Sauvanet A, Farges O. Seven hundred forty-seven hepatectomies in the 1990s: an update to evaluate the actual risk of liver resection. J Am Coll Surg. 2000;191:38–46.
Gomez D, Malik HZ, Bonney GK, et al. Steatosis predicts postoperative morbidity following hepatic resection for colorectal metastasis. Br J Surg. 2007;94:1395–402.
McCormack L, Petrowsky H, Jochum W, Furrer K, Clavien PA. Hepatic steatosis is a risk factor for postoperative complications after major hepatectomy: a matched case-control study. Ann Surg. 2007;245:923–30.
Hui AM, Kawasaki S, Makuuchi M, Nakayama J, Ikegami T, Miyagawa S. Liver injury following normothermic ischemia in steatotic rat liver. Hepatology. 1994;20:1287–93.
Selzner M, Rudiger HA, Sindram D, Madden J, Clavien PA. Mechanisms of ischemic injury are different in the steatotic and normal rat liver. Hepatology. 2000;32:1280–88.
Alexandre J, Nicco C, Chereau C, et al. Improvement of the therapeutic index of anticancer drugs by the superoxide dismutase mimic mangafodipir. J Natl Cancer Inst. 2006;98:236–44.
Kleiner DE, Brunt EM, Van Natta M, et al. Design and validation of a histological scoring system for nonalcoholic fatty liver disease. Hepatology. 2005;41:1313–21.
Day C, James O. Hepatic steatosis: innocent bystander or guilty party? Hepatology. 1998;27:1463–66.
Pessayre D, Berson A, Fromety B, et al. Mitochondria in steatohepatitis. Semin Liver Dis. 2001;21:57–69.
Yang S, Zhu H, Lane M, et al. Obesity increases sensitivity to endotoxin liver injury; implication for the pathogenesis of steatohepatitis. Proc Natl Acad Sci USA. 1997;94:2557–62.
Fernandez FG, Ritter J, Goodwin JW, Linehan DC, Hawkins WG, Strasberg SM. Effect of steatohepatitis associated with irinotecan or oxaliplatin on respectability of hepatic colorectal metastases. J Am Coll Surg. 2005;200:845–53.
Vauthey JN, Pawlik TM, Ribero D, et al. Chemotherapy regimen predicts steatohepatitis and an increase in 90-day mortality after surgery for hepatic colorectal metastases. J Clin Oncol. 2006;24:2065–72.
Rubbia-Brandt L, Audard V, Sartoretti P, et al. Severe hepatic sinusoidal obstruction associated with oxaliplatin-based chemotherapy in patients with metastatic colorectal cancer. Ann Oncol. 2004;15:460–6.
Karoui M, Penna C, Amin-Hashem M, et al. Influence of preoperative chemotherapy on the risk of major hepatectomy for colorectal liver metastases. Ann Surg. 2006;243:1–7.
Julie C, Lutz MP, Aust D, et al. Pathological analysis of hepatic injury after oxaliplatin-based neoadjuvant chemotherapy of colorectal cancer liver metastases: results of the EORTC intergroup phase III study 40983. J Clin Oncol 2007;25 [Abstract 241].
Aloia T, Sebagh M, Plasse M, et al. Liver histology and surgical outcomes after preoperative chemotherapy with fluorouracil plus oxaliplatin in colorectal cancer liver metastases. J Clin Oncol. 2006;24:4983–90.
Malik HZ, Farid S, Al-Mukthar A, et al. A critical appraisal of the role of neoadjuvant chemotherapy for colorectal liver metastases: a case-controlled study. Ann Surg Oncol. 2007;12:3519–26.
Welsh FK, Tilney HS, Tekkis PP, John TG, Rees M. Safe liver resection following chemotherapy for colorectal metastases is a matter of timing. Br J Cancer. 2007;96:1037–42.
Aloia T, Sebagh M, Plasse M, et al. Liver histology and surgical outcomes after preoperative chemotherapy with fluorouracil plus oxaliplatin in colorectal cancer liver metastases. J Clin Oncol. 2006;24:4983–90.
Kooby DA, Stockman J, Ben-Porat L, et al. Influence of transfusions on perioperative and long-term outcome in patients following hepatic resection for colorectal metastases. Colorectal metastases. Ann Surg. 2003;237:860–70.
Doria MI Jr, Shepard KV, Levin B, Riddell RH. Liver pathology following hepatic arterial infusion chemotherapy. Hepatic toxicity with FUDR. Cancer. 1986;58:855–61.
Chang AE, Schneider PD, Sugarbaker PH, et al. A prospective randomized trial of regional versus systemic continuous 5-fluorodoxyuridine chemotherapy in the treatment of colorectal liver metastases. Ann Surg. 1987;206:685–93.
Hohn DC, Stagg RJ, Friedman MA, et al. A randomized trial of continuous intravenous versus hepatic intraarterial floxuridine in patients with colorectal cancer metastatic to the liver: the northern California oncology group trial. J Clin Oncol. 1989;7:1646–54.
Kemeny M, Goldberg D, Beatty D, et al. Results of a prospective randomized trial of continuous regional chemotherapy and hepatic resection as treatment of hepatic metastases from colorectal primaries. Cancer. 1986;57:492–8.
Kemeny N, Daly J, Reichman B, et al. Intrahepatic or systemic infusion fluorodeoxyuridine in patients with liver metastases from colorectal carcinoma. A randomized trial. Ann Intern Med. 1987;107:459–65.
Bergmann JF, Rougier P, Liguory C, Zafrani ES, Metreau JM, Dhumeaux D. Sclerosing cholangitis after chemotherapy by continuous hepatic intra-arterial infusion of fluorodeoxyuridine. Gastroenterol Clin Biol. 1986;10:177–9.
Hohn DC, Stagg RJ, Friedman MA, et al. A randomized trial of continuous intravenous versus hepatic intraarterial floxuridine in patients with colorectal cancer metastatic to the liver: the northern California oncology group trial. J Clin Oncol. 1989;7:1646–1654.
Meric F, Patt YZ, Curley SA, et al. Surgery after downstaging of unresectable hepatic tumors with intraarterial chemotherapy. Ann Surg Oncol. 2000;7:490–5.
Elias D, Lasser P, Rougier P, Ducreux M, Bognel C, Roche A. Frequency, technical aspects, results, and indications of major hepatectomy after prolonged intra-arterial hepatic chemotherapy for initially unresectable hepatic tumors. J Am Coll Surg. 1995;180:213–9.
Clancy TE, Dixon E, Perlis R, Sutherland FR, Zinner MJ. Hepatic arterial infusion after curative resection of colorectal cancer metastases: a meta-analysis of prospective clinical trials. J Gastrointest Surg. 2005;9:198–206.
Martin JK, O’Connell MJ, Wieand HS, et al. Intra-arterial floxuridine versus systemic fluorouracil for hepatic metastases from colorectal cancer. Arch Surg. 1990;125:1022–7.
Skitzki JJ, Chang AE. Hepatic artery chemotherapy for colorectal liver metastases: technical considerations and review of clinical trials. Surg Oncol. 2002;11:123–35.
Onaitis M, Morse M, Hurwitz H, et al. Adjuvant hepatic arterial chemotherapy following metastasectomy in patients with isolated liver metastases. Ann Surg. 2003;237:782–8.
Link KH, Pillasch J, Formentini A, Sunelaitis E, Leder G, Safi F, et al. Downstaging by regional chemotherapy of non-resectable isolated colorectal liver metastases. Eur J Surg Oncol. 1999;25:381–8.
Bilchik AJ. Arterial chemotherapy as adjuvant and palliative treatment of hepatic colorectal metastases: an update. Surg Oncol Clin N Am. 2003;12:193–210.
Bajetta E, Buzzoni A, Giardini A, Bonadonna G. Liver assessment in women receiving adjuvant CMF chemotherapy. Tumori. 1981;67:27–30.
Kalowitz N, Aw TY, Simon FJ, Stolz A. Drug-induced hepatotoxicity. Ann Intem Med. 1986;104:826–39.
Perry M. Hepatotoxicity of chemotherapeutic agents. Semin Oncol. 1982;9:65–74.
Schein PS, Winokur SH. Immunosuppressive and cytotoxic chemotherapy: long-term complications. Ann Intem Med. 1975;82:84–95.
Torzili G, Makuuchi M, Inoue K, et al. No-mortality liver resection for hepatocellular carcinoma in cirrhotic and non-cirrhotic patients. Arch Surg. 1999;134:984–92.
Wu J, Ishikawa N, Takeda T, et al. The functional hepatic volume assessed by 99mTc-GSA hepatic scintigraphy. Ann Nucl Med. 1995;9:229–35.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Khan, A.Z., Morris-Stiff, G. & Makuuchi, M. Patterns of chemotherapy-induced hepatic injury and their implications for patients undergoing liver resection for colorectal liver metastases. J Hepatobiliary Pancreat Surg 16, 137–144 (2009). https://doi.org/10.1007/s00534-008-0016-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00534-008-0016-z