Abstract
Purpose
The treatment of splenic flexural colon cancer is not standardized because the lymphatic drainage is variable. The aim of this study is to evaluate the lymph flow at the splenic flexure.
Methods
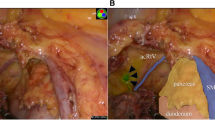
From July 2013 to January 2016, consecutive patients of the splenic flexural colon cancer with a preoperative diagnosis of N0 who underwent laparoscopic surgery were enrolled. Primary outcome is frequency of the direction of lymph flow from splenic flexure. We injected indocyanine green (2.5 mg) into the submucosal layer around the tumor and observed lymph flow using the laparoscopic near-infrared camera system in 30 min after injection.
Results
Thirty-one patients were enrolled in this study. The lymph flow was visualized in 31 patients (100 %) without any complications. No case exhibited lymph flow in both the left colic artery (LCA) and left branch of the middle colic artery (lt-MCA) areas. There were 19 cases (61.3 %) with lymph flow directed to the area of the root of the inferior mesenteric vein (IMV), regardless of the presence of the left accessory aberrant colic artery. Lymph node metastases were observed in six cases (19.4 %), and all of the involved lymph nodes existed in lymph flow areas determined by real-time indocyanine green fluorescence imaging.
Conclusions
The findings of the lymph flow pattern of splenic flexure suggest that lymph node dissection at the root of the IMV area is important, and it may be not necessary to ligate both the lt-MCA and LCA, at least in cases without widespread lymph node metastases.




Similar content being viewed by others
References
The Clinical Outcomes of Surgical Therapy Study Group (2004) A comparison of laparoscopically assisted and open colectomy for colon cancer. N Engl J Med 350(20):2050–2059
Guillou PJ, Quirke P, Thorpe H, Walker J, Jayne DG, Smith AM, Heath RM, Brown JM (2005) Short-term endpoints of conventional versus laparoscopic-assisted surgery in patients with colorectal cancer (MRC CLASICC trial): multicentre, randomised controlled trial. Lancet 365(9472):1718–1726
Veldkamp R, Kuhry E, Hop WC, Jeekel J, Kazemier G, Bonjer HJ, Haglind E, Pahlman L, Cuesta MA, Msika S, Morino M, Lacy AM (2005) Laparoscopic surgery versus open surgery for colon cancer: short-term outcomes of a randomised trial. Lancet Oncol 6(7):477–484
Yamamoto S, Inomata M, Katayama H, Mizusawa J, Etoh T, Konishi F, Sugihara K, Watanabe M, Moriya Y, Kitano S (2014) Short-term surgical outcomes from a randomized controlled trial to evaluate laparoscopic and open D3 dissection for stage II/III colon cancer: Japan Clinical Oncology Group Study JCOG 0404. Ann Surg 260(1):23–30
Buunen M, Veldkamp R, Hop WC, Kuhry E, Jeekel J, Haglind E, Pahlman L, Cuesta MA, Msika S, Morino M, Lacy A, Bonjer HJ (2009) Survival after laparoscopic surgery versus open surgery for colon cancer: long-term outcome of a randomised clinical trial. Lancet Oncol 10(1):44–52
Jayne DG, Thorpe HC, Copeland J, Quirke P, Brown JM, Guillou PJ (2010) Five-year follow-up of the Medical Research Council CLASICC trial of laparoscopically assisted versus open surgery for colorectal cancer. Br J Surg 97(11):1638–1645
Lacy AM, Delgado S, Castells A, Prins HA, Arroyo V, Ibarzabal A, Pique JM (2008) The long-term results of a randomized clinical trial of laparoscopy-assisted versus open surgery for colon cancer. Ann Surg 248(1):1–7
West NP, Hohenberger W, Weber K, Perrakis A, Finan PJ, Quirke P (2010) Complete mesocolic excision with central vascular ligation produces an oncologically superior specimen compared with standard surgery for carcinoma of the colon. J Clin Oncol 28(2):272–278
West NP, Kobayashi H, Takahashi K, Perrakis A, Weber K, Hohenberger W, Sugihara K, Quirke P (2012) Understanding optimal colonic cancer surgery: comparison of Japanese D3 resection and European complete mesocolic excision with central vascular ligation. J Clin Oncol 30(15):1763–1769
Pisani Ceretti A, Maroni N, Sacchi M, Bona S, Angiolini MR, Bianchi P, Opocher E, Montorsi M (2015) Laparoscopic colonic resection for splenic flexure cancer: our experience. BMC Gastroenterol 15:76
Perrakis A, Weber K, Merkel S, Matzel K, Agaimy A, Gebbert C, Hohenberger W (2014) Lymph node metastasis of carcinomas of transverse colon including flexures. Consideration of the extramesocolic lymph node stations. Int J Color Dis 29(10):1223–1229
Nakagoe T, Sawai T, Tsuji T, Jibiki M, Ohbatake M, Nanashima A, Yamaguchi H, Yasutake T, Kurosaki N, Ayabe H, Ishikawa H (2001) Surgical treatment and subsequent outcome of patients with carcinoma of the splenic flexure. Surg Today 31(3):204–209
Nakagoe T, Sawa T, Tsuji T, Jibiki M, Nanashima A, Yamaguchi H, Yasutake T, Ayabe H, Ishikawa H (2000) Carcinoma of the splenic flexure: multivariate analysis of predictive factors for clinicopathological characteristics and outcome after surgery. J Gastroenterol 35(7):528–535
Steffen C, Bokey EL, Chapuis PH (1987) Carcinoma of the splenic flexure. Dis Colon rectum 30(11):872–874
Khafagy MM, Stearns MW Jr (1973) Carcinoma of the splenic flexure. Dis Colon rectum 16(6):504–507
Jamieson JK, Dobson JF (1909) The lymphatics of the colon. Proc R Soc Med 2(Surg Sect):149–174
Rusu MC, Vlad M, Voinea LM, Curca GC, Sisu AM (2008) Detailed anatomy of a left accessory aberrant colic artery. Surg Radiol Anat 30(7):595–599
Mieog JS, Troyan SL, Hutteman M, Donohoe KJ, van der Vorst JR, Stockdale A, Liefers GJ, Choi HS, Gibbs-Strauss SL, Putter H, Gioux S, Kuppen PJ, Ashitate Y, Lowik CW, Smit VT, Oketokoun R, Ngo LH, van de Velde CJ, Frangioni JV, Vahrmeijer AL (2011) Toward optimization of imaging system and lymphatic tracer for near-infrared fluorescent sentinel lymph node mapping in breast cancer. Ann Surg Oncol 18(9):2483–2491
Jafari MD, Lee KH, Halabi WJ, Mills SD, Carmichael JC, Stamos MJ, Pigazzi A (2013) The use of indocyanine green fluorescence to assess anastomotic perfusion during robotic assisted laparoscopic rectal surgery. Surg Endosc 27(8):3003–3008
Jafari MD, Wexner SD, Martz JE, McLemore EC, Margolin DA, Sherwinter DA, Lee SW, Senagore AJ, Phelan MJ, Stamos MJ (2015) Perfusion assessment in laparoscopic left-sided/anterior resection (PILLAR II): a multi-institutional study. J Am Coll Surg 220(1):82–92 e81
Kudszus S, Roesel C, Schachtrupp A, Hoer JJ (2010) Intraoperative laser fluorescence angiography in colorectal surgery: a noninvasive analysis to reduce the rate of anastomotic leakage. Langenbeck’s Arch Surg 395(8):1025–1030
Nishigori N, Koyama F, Nakagawa T, Nakamura S, Ueda T, Inoue T, Kawasaki K, Obara S, Nakamoto T, Fujii H, Nakajima Y (2016) Visualization of lymph/blood flow in laparoscopic colorectal cancer surgery by ICG fluorescence imaging (lap-IGFI. Ann Surg Oncol 23(Suppl 2):266–274
Sherwinter DA, Gallagher J, Donkar T (2013) Intra-operative transanal near infrared imaging of colorectal anastomotic perfusion: a feasibility study. Color Dis 15(1):91–96
Aldridge MC, Phillips RK, Hittinger R, Fry JS, Fielding LP (1986) Influence of tumour site on presentation, management and subsequent outcome in large bowel cancer. Br J Surg 73(8):663–670
Mori T, Kimura T, Kitajima M (2010) Skill accreditation system for laparoscopic gastroenterologic surgeons in Japan. Minim Invasive Ther Allied Technol 19(1):18–23
Griffiths JD (1956) Surgical anatomy of the blood supply of the distal colon. Ann R Coll Surg Engl 19(4):241–256
Korn JM, Tellez-Diaz A, Bartz-Kurycki M, Gastman B (2014) Indocyanine green SPY elite-assisted sentinel lymph node biopsy in cutaneous melanoma. Plast Reconstr Surg 133(4):914–922
Reuthebuch O, Haussler A, Genoni M, Tavakoli R, Odavic D, Kadner A, Turina M (2004) Novadaq SPY: intraoperative quality assessment in off-pump coronary artery bypass grafting. Chest 125(2):418–424
Nagata J, Fukunaga Y, Akiyoshi T, Konishi T, Fujimoto Y, Nagayama S, Yamamoto N, Ueno M (2016) Colonic marking with near-infrared, light-emitting, diode-activated indocyanine green for laparoscopic colorectal surgery. Dis Colon rectum 59(2):e14–e18
Cahill RA, Anderson M, Wang LM, Lindsey I, Cunningham C, Mortensen NJ (2012) Near-infrared (NIR) laparoscopy for intraoperative lymphatic road-mapping and sentinel node identification during definitive surgical resection of early-stage colorectal neoplasia. Surg Endosc 26(1):197–204
Doekhie FS, Peeters KC, Kuppen PJ, Mesker WE, Tanke HJ, Morreau H, van de Velde CJ, Tollenaar RA (2005) The feasibility and reliability of sentinel node mapping in colorectal cancer. Eur J Surg Oncol 31(8):854–862
Des Guetz G, Uzzan B, Nicolas P, Cucherat M, de Mestier P, Morere JF, Breau JL, Perret G (2007) Is sentinel lymph node mapping in colorectal cancer a future prognostic factor? A meta-analysis. World J Surg 31(6):1304–1312
van der Pas MH, Meijer S, Hoekstra OS, Riphagen II, de Vet HC, Knol DL, van Grieken NC, Meijerink WJ (2011) Sentinel-lymph-node procedure in colon and rectal cancer: a systematic review and meta-analysis. Lancet Oncol 12(6):540–550
Kelder W, Braat AE, Karrenbeld A, Grond JA, De Vries JE, Oosterhuis JW, Baas PC, Plukker JT (2007) The sentinel node procedure in colon carcinoma: a multi-centre study in The Netherlands. Int J Color Dis 22(12):1509–1514
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
This study protocol was approved by the Ethics Advisory Committee of Yokosuka Kyosai Hospital before initiation and was registered in the UMIN Clinical Trials Registry as UMIN-CTR000013047 [http://www.umin.ac.jp/ctr/index.htm]. The procedure used during this study was explained, and written informed consent was obtained from all of the patients.
Conflict of interest
The authors declare that they have no conflicts of interest.
Rights and permissions
About this article
Cite this article
Watanabe, J., Ota, M., Suwa, Y. et al. Evaluation of lymph flow patterns in splenic flexural colon cancers using laparoscopic real-time indocyanine green fluorescence imaging. Int J Colorectal Dis 32, 201–207 (2017). https://doi.org/10.1007/s00384-016-2669-4
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00384-016-2669-4