Abstract
Background
Human phosphatases of regenerating liver (PRLs) can induce cell growth, differentiation, and malignant transformation. In this study, we used specific polyclonal antibodies against PRLs to investigate their expression in colonic adenocarcinomas and its correlation with patient gender, age, tumor differentiation, localization, invasion, and metastasis.
Materials and methods
The polyclonal antibodies against PRL-1, PRL-2, and PRL-3 were produced and purified. The expression of PRLs in human colorectal carcinoma cell lines (SW480 and SW620) was examined by Western blotting. We also examined their expression in normal and pathologic tissues from the human colon. The tissues included 49 primary colonic adenocarcinomas, 14 cases with lymph node metastases, 15 colonic adenomas, and 12 normal colon samples. Hematoxylin and eosin staining, immunohistochemistry, and semiquantitative morphological analysis were used to evaluate the sections.
Results
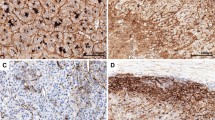
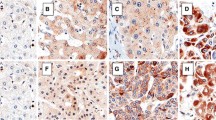
PRLs were widely expressed in SW480 and SW620. PRL-1, PRL-2, and PRL-3 were expressed, respectively, in 16, 10, and 16% of primary colonic adenocarcinomas. In contrast, PRLs were strongly expressed in all lymph node metastases. There were no significant correlations between the expression of PRLs and patient gender, age, tumor differentiation, depth of invasion, or localization of tumor within the different sections of the colon. PRLs were not expressed in normal colon tissues or in colonic adenomas. PRLs were mainly expressed in the cytoplasm and at the cytoplasmic membranes of the colonic adenocarcinoma cells as well as in the endothelial cells and the surrounding smooth muscle cells of larger vessels in the lymph node metastases.
Conclusion
Colonic adenocarcinoma cells have the ability to produce PRLs, which may relate to the lymph node metastasis of colonic adenocarcinoma.



Similar content being viewed by others
References
Jemal A, Murray T, Ward E (2005) Cancer statistics. CA Cancer J Clin 55:10–30
Saha S, Bardelli A, Buckhaults P (2001) A phosphatase associated with metastasis of colorectal cancer. Science 294(5545):1343–1346
Bardelli A, Saha S, Sager JA (2003) PRL-3 expression in metastatic cancers. Clin Cancer Res 9(15):5607–5615
Montagna M, Serova O, Sylla B (1995) A 100-kb physical and transcriptional map around the EDH17B2 gene: identification of three novel genes and a pseudogene of a human homologue of the rat PRL-1 tyrosine phosphatase. Hum Genet 96(5):532–538
Cates CA, Michael RL, Stayrook KR (1996) Prenylation of oncogenic human PTP(CAAX) protein tyrosine phosphatases. Cancer Lett 110(1–2):49–55
Diamond RH, Cressman DE, Laz TM (1994) PRL-1, a unique nuclear protein tyrosine phosphatase, affects cell growth. Mol Cell Biol 14(6):3752–3762
Zhao Z, Lee CC, Monckton DG (1996) Characterization and genomic mapping of genes and pseudogenes of a new human protein tyrosine phosphatase. Genomics 35(1):172–181
Carter DA (1998) Expression of a novel rat protein tyrosine phosphatase gene. Biochim Biophys Acta 1442(2–3):405–408
Haber BA, Mohn KL, Diamond RH (1993) Induction patterns of 70 genes during nine days after hepatectomy define the temporal course of liver regeneration. J Clin Invest 91(4):1319–1326
Mohn KL, Laz TM, Hsu JC (1991) The immediate-early growth response in regenerating liver and insulin-stimulated H-35 cells: comparison with serum-stimulated 3T3 cells and identification of 41 novel immediate-early genes. Mol Cell Biol 11(1):381–930
Diamond RH, Peters C, Jung SP (1996) Expression of PRL-1 nuclear PTPase is associated with proliferation in liver but with differentiation in intestine. Am J Physiol 271(1 Pt 1):G121–G129
Nigar F, Dhirendra PS, Toshimichi S (2000) Heparin’s roles in stabilizing, potentiating, and transporting LEDGF into the nucleus. Investig Ophthalmol Vis Sci 41:2648–2657
Erhard MH, Jungling A, Brendgen S (1992) Development of a direct and indirect chemiluminescence immunoassay for the detection of an organophosphorus compound. J Immunoass 13(2):273–287
Zeng Q, Si X, Horstmann H (2000) Prenylation-dependent association of protein-tyrosine phosphatases PRL-1, -2, and -3 with the plasma membrane and the early endosome. J Biol Chem 275(28):21444–21452
Johnston SR (2001) Farnesyl transferase inhibitors: a novel targeted therapy for cancer. Lancet Oncol 2(1):18–26
Peng LR, Li Y, Meng L (2004) Preparation and characterization of monoclonal antibody against protein tyrosine phosphatase PRL-3. Hybrid Hybridomics 23(1):23–27
Zeng Q, Dong JM, Guo K (2003) PRL-3 and PRL-1 promote cell migration, invasion, and metastasis. Cancer Res 63:2716–2722
Acknowledgment
We are grateful to Dr. Zhao Zhizhuang Joe for providing us with DE3pLysS E. coli. Furthermore, we thank Dr. Frederick William Orr (Professor, Department of Pathology, University of Manitoba, Canada) for facilitating the preparation of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Y. Wang and Z.-F. Li contributed equally to this work.
Rights and permissions
About this article
Cite this article
Wang, Y., Li, ZF., He, J. et al. Expression of the human phosphatases of regenerating liver (PRLs) in colonic adenocarcinoma and its correlation with lymph node metastasis. Int J Colorectal Dis 22, 1179–1184 (2007). https://doi.org/10.1007/s00384-007-0303-1
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00384-007-0303-1