Abstract
Purpose
Time to metastasis is often used as a surrogate parameter of treatment success in clinical trials for prostate cancer. However, it has not been shown that there is a clear correlation between time to metastasis and overall survival. Our objective was to evaluate the impact of time to metastasis on OS in patients with prostate cancer.
Methods
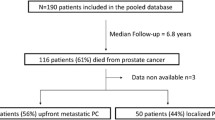
Between 2008 and 2015, 269 patients with mPCa were included in this retrospective study with a median follow-up of 7.1 years. Patients were divided into three groups: (1) Presentation with metastasis within three months of initial diagnosis (de-novo-M); (2) patients free of metastasis initially but developed metastasis more than 6 months prior to castration resistance (CSPC-M); (3) patients who developed metastasis within 6 months of becoming castration resistant or after (CRPC-M).
Results
There was a significant decrease in OS when metastases were present at diagnosis (median 6.39 years) compared to CRPC-M (19.07) and CSPC-M (18.19 years). De-novo-M and CSPC-M showed a longer OS from occurrence of metastasis to death when compared to CRPC-M, although reaching CRPC earlier. There was no difference in OS between the groups once castration resistance was reached. Time from initial diagnosis to metastasis and to CRPC was correlated with OS and remained important prognosticators in multivariate Cox-regression (p < 0.01 for both).
Conclusions
Time from diagnosis to CRPC (all patients) and time to metastasis (for CRPC-M and CSPC-M patients) are significant prognosticators of overall survival and are therefore valid surrogates in a study setting. Therefore, time to CRPC should be prolonged as long as possible.

Similar content being viewed by others
References
Society AC (2016) Cancer facts & figures 2016. American Cancer Society, Atlanta
Stephenson R (2005) Prostate cancer overdiagnosis and overtreatment: analysis of US mortality and SEER incidence. Trends in the PSA and pre-PSA eras. In: Klein EA (ed) Management of prostate cancer. Humana Press, Totowa (NJ), pp 3–13
Glass TR, Tangen CM, Crawford ED, Thompson I (2003) Metastatic carcinoma of the prostate: identifying prognostic groups using recursive partitioning. J Urol 169(1):164–169. https://doi.org/10.1097/01.ju.0000042482.18153.30
Gravis G, Boher JM, Fizazi K, Joly F, Priou F, Marino P, Latorzeff I, Delva R, Krakowski I, Laguerre B, Walz J, Rolland F, Theodore C, Deplanque G, Ferrero JM, Pouessel D, Mourey L, Beuzeboc P, Zanetta S, Habibian M, Berdah JF, Dauba J, Baciuchka M, Platini C, Linassier C, Labourey JL, Machiels JP, El Kouri C, Ravaud A, Suc E, Eymard JC, Hasbini A, Bousquet G, Soulie M, Oudard S (2015) Prognostic factors for survival in noncastrate metastatic prostate cancer: validation of the glass model and development of a novel simplified prognostic model. Eur Urol 68(2):196–204. https://doi.org/10.1016/j.eururo.2014.09.022
Halabi S, Small EJ, Kantoff PW, Kattan MW, Kaplan EB, Dawson NA, Levine EG, Blumenstein BA, Vogelzang NJ (2003) Prognostic model for predicting survival in men with hormone-refractory metastatic prostate cancer. J Clin Oncol 21(7):1232–1237
Smaletz O, Scher HI, Small EJ, Verbel DA, McMillan A, Regan K, Kelly WK, Kattan MW (2002) Nomogram for overall survival of patients with progressive metastatic prostate cancer after castration. J Clin Oncol 20(19):3972–3982
Armstrong AJ, Garrett-Mayer ES, Yang YC, de Wit R, Tannock IF, Eisenberger M (2007) A contemporary prognostic nomogram for men with hormone-refractory metastatic prostate cancer: a TAX327 study analysis. Clin Cancer Res 13(21):6396–6403. https://doi.org/10.1158/1078-0432.CCR-07-1036
Safety and Efficacy Study of Enzalutamide in Patients With Nonmetastatic Castration-Resistant Prostate Cancer (PROSPER)
A Study of Apalutamide (ARN-509) in Men With Non-Metastatic Castration-Resistant Prostate Cancer (SPARTAN)
Mottet N, Bellmunt J, Briers, E, van den Bergh RCN, Bolla M, van Casteren NJ, Cornford P, Culine S, Joniau S, Lam T, Mason MD, Matveev V, van der Poel THvdK H, Rouvière O, Wiegel T (2015) Guidelines on Prostate Cancer. https://uroweb.org/guideline/prostate-cancer/. Mar 2015
Patrikidou A, Loriot Y, Eymard JC, Albiges L, Massard C, Ileana E, Di Palma M, Escudier B, Fizazi K (2014) Who dies from prostate cancer? Prostate Cancer Prostatic Dis 17(4):348–352. https://doi.org/10.1038/pcan.2014.35
Patrikidou A, Brureau L, Casenave J, Albiges L, Di Palma M, Patard JJ, Baumert H, Blanchard P, Bossi A, Kitikidou K, Massard C, Fizazi K, Blanchet P, Loriot Y (2015) Locoregional symptoms in patients with de novo metastatic prostate cancer: morbidity, management, and disease outcome. Urol Oncol 33(5):202–e209-217. https://doi.org/10.1016/j.urolonc.2015.01.022
Sweeney CJ, Chen YH, Carducci M, Liu G, Jarrard DF, Eisenberger M, Wong YN, Hahn N, Kohli M, Cooney MM, Dreicer R, Vogelzang NJ, Picus J, Shevrin D, Hussain M, Garcia JA, DiPaola RS (2015) Chemohormonal therapy in metastatic hormone-sensitive prostate cancer. N Engl J Med 373(8):737–746. https://doi.org/10.1056/NEJMoa1503747
James ND, Sydes MR, Clarke NW, Mason MD, Dearnaley DP, Spears MR, Ritchie AW, Parker CC, Russell JM, Attard G, de Bono J, Cross W, Jones RJ, Thalmann G, Amos C, Matheson D, Millman R, Alzouebi M, Beesley S, Birtle AJ, Brock S, Cathomas R, Chakraborti P, Chowdhury S, Cook A, Elliott T, Gale J, Gibbs S, Graham JD, Hetherington J, Hughes R, Laing R, McKinna F, McLaren DB, O’Sullivan JM, Parikh O, Peedell C, Protheroe A, Robinson AJ, Srihari N, Srinivasan R, Staffurth J, Sundar S, Tolan S, Tsang D, Wagstaff J, Parmar MK (2016) Addition of docetaxel, zoledronic acid, or both to first-line long-term hormone therapy in prostate cancer (STAMPEDE): survival results from an adaptive, multiarm, multistage, platform randomised controlled trial. Lancet 387(10024):1163–1177. https://doi.org/10.1016/S0140-6736(15)01037-5
Fizazi K, Tran N, Fein L, Matsubara N, Rodriguez-Antolin A, Alekseev BY, Ozguroglu M, Ye D, Feyerabend S, Protheroe A, De Porre P, Kheoh T, Park YC, Todd MB, Chi KN, Investigators L (2017) Abiraterone plus prednisone in metastatic, castration-sensitive prostate cancer. N Engl J Med 377(4):352–360. https://doi.org/10.1056/NEJMoa1704174
James ND, de Bono JS, Spears MR, Clarke NW, Mason MD, Dearnaley DP, Ritchie AWS, Amos CL, Gilson C, Jones RJ, Matheson D, Millman R, Attard G, Chowdhury S, Cross WR, Gillessen S, Parker CC, Russell JM, Berthold DR, Brawley C, Adab F, Aung S, Birtle AJ, Bowen J, Brock S, Chakraborti P, Ferguson C, Gale J, Gray E, Hingorani M, Hoskin PJ, Lester JF, Malik ZI, McKinna F, McPhail N, Money-Kyrle J, O’Sullivan J, Parikh O, Protheroe A, Robinson A, Srihari NN, Thomas C, Wagstaff J, Wylie J, Zarkar A, Parmar MKB, Sydes MR, Investigators S (2017) Abiraterone for prostate cancer not previously treated with hormone therapy. N Engl J Med 377(4):338–351. https://doi.org/10.1056/NEJMoa1702900
James ND, Spears MR, Clarke NW, Dearnaley DP, De Bono JS, Gale J, Hetherington J, Hoskin PJ, Jones RJ, Laing R, Lester JF, McLaren D, Parker CC, Parmar MK, Ritchie AW, Russell JM, Strebel RT, Thalmann GN, Mason MD, Sydes MR (2015) Survival with newly diagnosed metastatic prostate cancer in the “docetaxel era”: data from 917 patients in the control arm of the STAMPEDE trial (MRC PR08, CRUK/06/019). Eur Urol 67(6):1028–1038. https://doi.org/10.1016/j.eururo.2014.09.032
Wallis CJD, Klaassen Z, Bhindi B, Goldberg H, Chandrasekar T, Farrell AM, Boorjian SA, Kulkarni GS, Karnes RJ, Satkunasivam R (2017) Comparison of abiraterone acetate and docetaxel with androgen deprivation therapy in high-risk and metastatic hormone-naive prostate cancer: a systematic review and network meta-analysis. Eur Urol. https://doi.org/10.1016/j.eururo.2017.10.002
Qin XJ, Ma CG, Ye DW, Yao XD, Zhang SL, Dai B, Zhang HL, Shen YJ, Zhu Y, Zhu YP, Shi GH, Xiao WJ, Lin GW, Swanson GP (2012) Tumor cytoreduction results in better response to androgen ablation–a preliminary report of palliative transurethral resection of the prostate in metastatic hormone sensitive prostate cancer. Urol Oncol 30(2):145–149. https://doi.org/10.1016/j.urolonc.2010.02.010
Swanson G, Thompson I, Basler J, Crawford ED (2006) Metastatic prostate cancer-does treatment of the primary tumor matter? J Urol 176(4 Pt 1):1292–1298. https://doi.org/10.1016/j.juro.2006.06.069
Culp SH, Schellhammer PF, Williams MB (2014) Might men diagnosed with metastatic prostate cancer benefit from definitive treatment of the primary tumor? A SEER-based study. Eur Urol 65(6):1058–1066. https://doi.org/10.1016/j.eururo.2013.11.012
Fossati N, Trinh QD, Sammon J, Sood A, Larcher A, Sun M, Karakiewicz P, Guazzoni G, Montorsi F, Briganti A, Menon M, Abdollah F (2015) Identifying optimal candidates for local treatment of the primary tumor among patients diagnosed with metastatic prostate cancer: a SEER-based study. Eur Urol 67(1):3–6. https://doi.org/10.1016/j.eururo.2014.08.056
Acknowledgements
Dr. Frees held a scholarship from the “Deutsche Forschungsgemeinschaft” (DFG).
Author information
Authors and Affiliations
Contributions
S. Frees Protocol/project development, Data collection or management, Data analysis, Manuscript writing/editing. S. Akamatsu Protocol/project development, Data collection or management, Manuscript writing/editing. S. Bidnur Manuscript writing/editing. D. Khalaf Data collection or management. C. Chavez-Munoz Data analysis, Manuscript writing/editing. W.Struss Data collection or management, Manuscript writing/editing. B. J Eigl Protocol/project development, Manuscript writing/editing. M. Gleave Protocol/project development, Manuscript writing/editing. K. N. Chi Protocol/project development, Manuscript writing/editing. A. I. So Protocol/project development, Data analysis, Manuscript writing/editing.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Statement of human rights
For this type of study formal consent is not required.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Electronic supplementary material
Below is the link to the electronic supplementary material.
345_2018_2236_MOESM1_ESM.pdf
Supplementary material 1 (PDF 465 kb). Supplementary Fig. 2: Survival from time of metastasis. Kaplan–Meier analysis of the overall survival of patients from time of metastasis. Supplementary Fig. 3: Time from diagnosis to metastasis (CRPC-M vs CSPC-M). Kaplan–Meier analysis of the time from diagnosis to metastasis. Supplementary Fig. 4: a Pearson correlation of time from diagnosis to metastasis and time from diagnosis to death. b Pearson Correlation of time from diagnosis to CRPC and time from diagnosis to death
345_2018_2236_MOESM2_ESM.pdf
Supplementary material 2 (PDF 448 kb). Supplementary Fig. 5: Time from CRPC to death. Kaplan–Meier analysis of the overall survival of patients from time of becoming castration resistant. Supplementary Fig. 6:De- novo-M compared to all other patients divided into groups according to time to metastasis. Kaplan–Meier analysis of the overall survival of patients comparing de-novo-metastasis and groups of different time intervals of metastasis
345_2018_2236_MOESM3_ESM.pdf
Supplementary material 3 (PDF 331 kb). Supplementary Fig. 1: Schematic timeline of a typical course of patients with prostate cancer and the division used in this paper according to the time point of metastasis. Above: Initial non-metastatic prostate cancer, that is treated with an initial therapy, mostly in a curative intent, then biochemical failure which is typically treated with Androgen deprivation therapy (ADT). Patients become castration resistant followed by a secondary ADT or chemotherapy and finally ending in the death of the patient. Below: Patients with initially metastatic prostate cancer receive ADT and once becoming castration resistant chemotherapy or secondary ADT. De novo-M: Patients that presented with clinical evidence of metastasis within 3 months of initial diagnosis; CSPC-M (castration sensitive prostate cancer with metastasis): Patients who were free of metastasis initially but developed metastasis more than 6 months prior to castration resistance; CRPC-M (castration resistant prostate cancer with metastasis) patients who developed metastasis within 6 months of becoming castration resistant or after. Abbreviations: CRPC: castration resistant prostate cancer
345_2018_2236_MOESM4_ESM.docx
Supplementary material 4 (DOCX 142 kb). Supplementary Table 1: Multivariate Cox- regression: A: Prognosticators influencing OS for patients with CRPC-M and CSPC-M; B: Prognosticators influencing OS for all patients
Rights and permissions
About this article
Cite this article
Frees, S., Akamatsu, S., Bidnur, S. et al. The impact of time to metastasis on overall survival in patients with prostate cancer. World J Urol 36, 1039–1046 (2018). https://doi.org/10.1007/s00345-018-2236-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00345-018-2236-4