Abstract
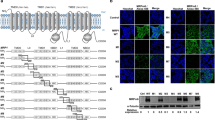
The present study was undertaken to identify what regulates intracellular cisplatin (CDDP) accumulation and what changes in membrane fraction of CDDDP-resistant cell line. The CDDP-resistant rat hepatoma cell line, H4-II-E/CDDP, shows a significant decrease in intracellular platinum accumulation compared with parental H4-II-E cells, although there was no difference in the efflux of CDDP between these two cell lines. In this study, we examined the contribution of functional change in active transport to the CDDP resistance of H4-II-E/CDDP cells. Compared with the resistant cells, platinum accumulation in the parental cells was clearly decreased by low temperature or ATP depletion. In addition, the Na+, K+-ATPase inhibitor ouabain and the K+ channel inhibitor tetraethylammonium decreased platinum accumulation in parental cells but did not change the accumulation in resistant cells. Amphotericin B, an antifungal agent, increased the intracellular platinum accumulation in resistant cells to the same level as in parent cells. Western blot analysis demonstrated that the Na+, K+-ATPase α1 subunit was reduced in resistant cells compared with the parental cells, although there was no difference in the expression of the β1 subunit between the two cell lines. Furthermore, the Na+, K+-ATPase α1 subunit of H4-II-E was decreased following a 24-h exposure to CDDP. These results suggest that Na+, K+-ATPase-dependent active transport of CDDP does not occur in resistant cells, and, furthermore, our findings provide the first evidence that the Na+, K+-ATPase α1 subunit plays an important role in the transport of CDDP.







Similar content being viewed by others
References
Perez RP (1998) Cellular and molecular determinants of cisplatin resistance. Eur J Cancer 34:1535–1542
Siddik ZH (2003) Cisplatin: mode of cytotoxic action and molecular basis of resistance. Oncogene 22:7265–7279
Fuertes MA, Alonso C, Perez JM (2003) Biochemical modulation of Cisplatin mechanisms of action: enhancement of antitumor activity and circumvention of drug resistance. Chem Rev 103:645–662
Kelland LR, Sharp SY, O‘Neill CF, Raynaud FI, Beale PJ, Judson IR (1999) Mini-review: discovery and development of platinum complexes designed to circumvent cisplatin resistance. J Inorg Biochem 77:111–115
Gately DP, Howell SB (1993) Cellular accumulation of the anticancer agent cisplatin: a review. Br J Cancer 67:1171–1176
Mann SC, Andrews PA, Howell SB (1988) Comparison of lipid content, surface membrane fluidity, and temperature dependence of cis-diamminedichloroplatinum(II) accumulation in sensitive and resistant human ovarian carcinoma cells. Anticancer Res 8:1211–1215
Andrews PA, Mann SC, Huynh HH, Albright KD (1991) Role of the Na+, K+-adenosine triphosphatase in the accumulation of cis-diamminedichloroplatinum(II) in human ovarian carcinoma cells. Cancer Res 51:3677–3681
Andrews PA, Albright KD (1992) Mitochondrial defects in cis-diamminedichloroplatinum(II)-resistant human ovarian carcinoma cells. Cancer Res 52:1895–1901
Komatsu M, Sumizawa T, Mutoh M, Chen Z-S, Terada K, Furukawa T, Yang X-L, Gao H, Miura N, Sugiyama T, Akiyama S (2000) Copper-transporting P-type adenosine triphosphatase (ATP7B) is associated with cisplatin resistance. Cancer Res 60:1312–1316
Ishida S, Lee J, Thiele DJ, Herskowitz I (2002) Uptake of the anticancer drug cisplatin mediated by the copper transporter Ctr1 in yeast and mammals. Proc Natl Acad Sci USA 99:14298–14302
Kishimoto S, Miyazawa K, Terakawa Y, Ashikari H, Ohtani A, Fukushima S, Takeuchi Y (2000) Cytotoxicity of cis-[((1R,2R)-1,2-cyclohexanediamine-N,N’)bis(myristato)]-platinum (II) suspended in Lipiodol in a newly established cisplatin-resistant rat hepatoma cell line. Jpn J Cancer Res 91:1326–1332
Dornish JM, Pettersen EO (1985) Protection from cis-dichlorodiammineplatinum-induced cell inactivation by aldehydes involves cell membrane amino groups. Cancer Lett 29:235–243
Loh SY, Mistry P, Kelland LR, Abel G, Harrap KR (1992) Reduced drug accumulation as a major mechanism of acquired resistance to cisplatin in a human ovarian carcinoma cell line: circumvention studies using novel platinum (II) and (IV) ammine/amine complexes. Br J Cancer 66:1109–1115
Bando T, Fujimura M, Kasahara K, Matsuda T (1998) Significance of Na+, K+-ATPase on intracellular accumulation of cis-diamminedichloroplatinum(II) in human non-small-cell but not in small-cell lung cancer cell lines. Anticancer Res 18:1085–1089
Lizuka N, Miyamoto K, Tangoku A, Hayashi H, Hazama S, Yoshino S, Yoshimura K, Hirose K, Yoshida H, Oka M (2000) Downregulation of intracellular nm23-H1 prevents cisplatin-induced DNA damage in oesophageal cancer cells: possible association with Na+, K+-ATPase. Br J Cancer 83:1209–1215
Katano K, Kondo A, Safaei R, Holzer A, Samimi G, Mishima M, Kuo YM, Rochdi M, Howell SB (2002) Acquisition of resistance to cisplatin is accompanied by changes in the cellular pharmacology of copper. Cancer Res 62:6559–6565
Lin X, Okuda T, Holzer A, Howell SB (2002) The copper transporter CTR1 regulates cisplatin uptake in Saccharomyces cerevisiae. Mol Pharmacol 62:1154–1159
Beretta GL, Gatti L, Tinelli S, Corna E, Colangelo D, Zunino F, Perego P (2004) Cellular pharmacology of cisplatin in relation to the expression of human copper transporter CTR1 in different pairs of cisplatin-sensitive and -resistant cells. Biochem Pharmacol 68:283–291
Holzer AK, Samimi G, Katano K, Naerdemann W, Lin X, Safaei R, Howell SB (2004) The copper influx transporter hCTR1 regulates the uptake of cisplatin in human ovarian carcinoma cells. Mol Pharmacol 66:817–823
Samimi G, Katano K, Holzer AK, Safaei R, Howell SB (2004) Modulation of the cellular pharmacology of cisplatin and its analogs by the copper exporters ATP7A and ATP7B. Mol Pharmacol 66:25–32
Lingrel JB, Kuntzweiler T (1994) Na+, K+-ATPase. J Biol Chem 269:19659–19662
Rose AM, Valdes R Jr (1994) Understanding the sodium pump and its relevance to disease. Clin Chem 40:1674–1685
Geering K (1990) Subunit assembly and functional maturation of Na,K-ATPase. J Membr Biol 115:109–121
McDonough AA, Geering K, Farley RA (1990) The sodium pump needs its beta subunit. FASEB J 4:1598–1605
Takeyasu K, Tamkun MM, Renaud KJ, Fambrough DM (1988) Ouabain-sensitive (Na+, K+)-ATPase activity expressed in mouse L cells by transfection with DNA encoding the alpha-subunit of an avian sodium pump. J Biol Chem 263:4347–4354
Andrews PA, Velury S, Mann SC, Howell SB (1988) cis-Diamminedichloroplatinum(II) accumulation in sensitive and resistant human ovarian carcinoma cells. Cancer Res 48:68–73
Sharp SY, Rogers PM, Kelland LR (1995) Transport of cisplatin and bis-acetato-ammine-dichlorocyclohexylamine Platinum (IV) (JM216) in human ovarian carcinoma cell lines: identification of a plasma membrane protein associated with cisplatin resistance. Clin Cancer Res 1:981–989
Komuro Y, Udagawa Y, Susumu N, Aoki D, Kubota T, Nozawa S (2001) Paclitaxel and SN-38 overcome cisplatin resistance of ovarian cancer cell lines by down-regulating the influx and efflux system of cisplatin. Jpn J Cancer Res 92:1242–1250
Kolb RJ, Ghazi AM, Barfuss DW (2003) Inhibition of basolateral transport and cellular accumulation of cDDP and N-acetyl-L-cysteine-cDDP by TEA and PAH in the renal proximal tubule. Cancer Chemother Pharmacol 51:132–138
Kikkawa F, Kojima M, Oguchi H, Maeda O, Ishikawa H, Tamakoshi K, Mizuno K, Kawai M, Suganuma N, Tomoda Y (1993) Potentiating effect of amphotericin B on five platinum anticancer drugs in human cis-diamminedichloroplatinum (II) sensitive and resistant ovarian carcinoma cells. Anticancer Res 13:891–896
Morikage T, Bungo M, Inomata M, Yoshida M, Ohmori T, Fujiwara Y, Nishio K, Saijo N (1991) Reversal of cisplatin resistance with amphotericin B in a non-small cell lung cancer cell line. Jpn J Cancer Res 82:747–751
Delamere NA, Dean WL, Stidam JM, Moseley AE (1996) Influence of amphotericin B on the sodium pump of porcine lens epithelium. Am J Physiol 270:C465–C473
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kishimoto, S., Kawazoe, Y., Ikeno, M. et al. Role of Na+, K+-ATPase α1 subunit in the intracellular accumulation of cisplatin. Cancer Chemother Pharmacol 57, 84–90 (2006). https://doi.org/10.1007/s00280-005-0003-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00280-005-0003-x