Abstract
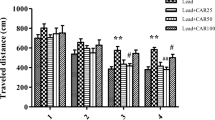
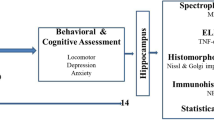
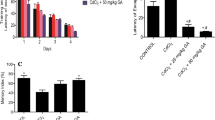
Increasing evidence suggests that lead (Pb) produces impairments partly through oxidative stress. Though many researchers have investigated protective effect of some antioxidant nutrients against Pb toxicity, little information is available about the effect of antioxidants on Pb-induced impairment of synaptic plasticity. Quercetin, a strong antioxidant and radical scavenger, is the representative natural flavonoid molecule abundant in fruits and vegetables. Previous studies have found that quercetin was neuroprotective in many cases. This study was designed to evaluate the effect of quercetin on chronic Pb exposure-induced impairment of synaptic plasticity in adult rat dentate gyrus (DG) area in vivo. The input/output (I/O) functions, paired-pulse reactions (PPR), excitatory postsynaptic potential (EPSP), and population spike (PS) amplitude were measured in the DG area of different groups of rats in response to stimulation applied to the lateral perforant path. The results showed that the depressed I/O, PPR, and long-term potentiation (LTP) of Pb-exposed group were significantly increased by quercetin treatment. In addition, hippocampal Pb concentration was partially reduced after quercetin treatment. These findings suggest that quercetin treatment could relieve chronic Pb exposure-induced impairment of synaptic plasticity and might be a potential therapeutic intervention to cure cognitive deficits induced by Pb.






Similar content being viewed by others
Abbreviations
- control + Que:
-
control with quercetin treatment
- CREB:
-
cyclic-AMP response element binding protein
- DG:
-
dentate gyrus
- EPSP:
-
excitatory postsynaptic potential
- I/O:
-
input/output functions
- IPI:
-
interpulse interval
- LTP:
-
long-term potentiation
- Pb + Que:
-
chronic lead exposed with quercetin treatment
- PPF:
-
paired-pulse facilitation
- PPR:
-
paired-pulse reactions
- PS:
-
population spike
- ROS:
-
reactive oxygen species
References
Adonaylo VN, Oteiza PI (1999) Lead intoxication: antioxidant defenses and oxidative damage in rat brain. Toxicology 135:77–85
Altmann L, Weinsberg F, Sveinsson K, Lilienthal H, Wiegand H, Winneke G (1993) Impairment of long-term potentiation and learning following chronic lead exposure. Toxicol Lett 66:105–112
Aykin-Burns N, Laegeler A, Kellogg G, Ercal N (2003) Oxidative effects of lead in young and adult Fisher 344 rats. Arch Environ Contam Toxicol 44:417–420
Batra N, Nehru B, Bansal MP (1998) The effect of zinc supplementation on the effects of lead on the rat testis. Reprod Toxicol 12:535–540
Bellinger D, Sloman J, Leviton A, Rabinowitz M, Needleman HL, Waternaux C (1991) Low-level lead exposure and children’s cognitive function in the preschool years. Pediatrics 87:219–227
Bennet C, Bettaiya R, Rajanna S, Baker L, Yallapragada PR, Brice JJ, White SL, Bokara KK (2007) Region specific increase in the antioxidant enzymes and lipid peroxidation products in the brain of rats exposed to lead. Free Radic Res 41:267–273
Bliss TV, Collingridge GL (1993) A synaptic model of memory: long-term potentiation in the hippocampus. Nature 361:31–39
Bourtchuladze R, Frenguelli B, Blendy J, Cioffi D, Schutz G, Silva AJ (1994) Deficient long-term memory in mice with a targeted mutation of the cAMP-responsive element-binding protein. Cell 79:59–68
Chaurasia SS, Kar A (1997) Protective effects of vitamin E against lead-induced deterioration of membrane associated type-I iodothyronine 5¢-monodeiodinase (5¢D-I) activity in male mice. Toxicology 124:203–209
Cho JY, Kim IS, Jang YH, Kim AR, Lee SR (2006) Protective effect of quercetin, a natural flavonoid against neuronal damage after transient global cerebral ischemia. Neurosci Lett 404:330–335
Cornard JP, Dangleterre L, Lapouge C (2005) Computational and spectroscopic characterization of the molecular and electronic structure of the Pb(II)–quercetin complex. J Phys Chem A Mol Spectrosc Kinet Environ Gen Theory 109:10044–10051
Cory-Slechta DA (1995) Relationships between lead-induced learning impairments and changes in dopaminergic, cholinergic, and glutamatergic neurotransmitter system functions. Annu Rev Pharmacol Toxicol 35:391–415
Croft KD (1998) The chemistry and biological effects of flavonoids and phenolic acids. Ann N Y Acad Sci 854:435–442
Deisseroth K, Bito H, Tsien RW (1996) Signaling from synapse to nucleus: postsynaptic CREB phosphorylation during multiple forms of hippocampal synaptic plasticity. Neuron 16:89–101
Dhawan M, Flora SJ, Tandon SK (1989) Preventive and therapeutic role of vitamin E in chronic plumbism. Biomed Environ Sci 2:335–340
Ding Y, Gonick HC, Vaziri ND (2000) Lead promotes hydroxyl radical generation and lipid peroxidation in cultured aortic endothelial cells. Am J Hypertens 13:552–555
Dok-Go H, Lee KH, Kim HJ, Lee EH, Lee J, Song YS, Lee YH, Jin C, Lee YS, Cho J (2003) Neuroprotective effects of antioxidative flavonoids, quercetin, (+)-dihydroquercetin and quercetin 3-methyl ether, isolated from Opuntia ficus-indica var. saboten. Brain Res 965:130–136
Geleijnse JM, Launer LJ, Van der Kuip DA, Hofman A, Witteman JC (2002) Inverse association of tea and flavonoid intakes with incident myocardial infarction: the Rotterdam Study. Am J Clin Nutr 75:880–886
Gilbert ME, Mack CM, Lasley SM (1996) Chronic developmental lead exposure increases the threshold for long-term potentiation in rat dentate gyrus in vivo. Brain Res 736:118–124
Gilbert ME, Mack CM, Lasley SM (1999) The influence of developmental period of lead exposure on long-term potentiation in the adult rat dentate gyrus in vivo. Neurotoxicology 20:57–69
Gurer H, Ercal N (2000) Can antioxidants be beneficial in the treatment of lead poisoning? Free Radic Biol Med 29:927–945
Harwood M, Danielewska-Nikiel B, Borzelleca JF, Flamm GW, Williams GM, Lines TC (2007) A critical review of the data related to the safety of quercetin and lack of evidence of in vivo toxicity, including lack of genotoxic/carcinogenic properties. Food Chem Toxicol 45:2179–2205
Hsu PC, Guo YL (2002) Antioxidant nutrients and lead toxicity. Toxicology 180:33–44
Hsu PC, Liu MY, Hsu CC, Chen LY, Leon Guo Y (1997) Lead exposure causes generation of reactive oxygen species and functional impairment in rat sperm. Toxicology 122:133–143
Impey S, Mark M, Villacres EC, Poser S, Chavkin C, Storm DR (1996) Induction of CRE-mediated gene expression by stimuli that generate long-lasting LTP in area CA1 of the hippocampus. Neuron 16:973–982
Klaassen CD (1990) Heavy metals and heavy metal antagonists. In: Gilman AG, Rall TW, Nies AS, Taylor P (eds) Goodman & Gilman’s the pharmacological basis of therapeutics. New York, Peragamon
Knekt P, Jarvinen R, Seppanen R, Hellovaara M, Teppo L, Pukkala E, Aromaa A (1997) Dietary flavonoids and the risk of lung cancer and other malignant neoplasms. Am J Epidemiol 146:223–230
Lasley SM, Gilbert ME (2000) Glutamatergic components underlying lead-induced impairments in hippocampal synaptic plasticity. Neurotoxicology 21:1057–1068
Liu J, Yu H, Ning X (2006) Effect of quercetin on chronic enhancement of spatial learning and memory of mice. Sci China C Life Sci 49:583–590
Lu J, Zheng YL, Luo L, Wu DM, Sun DX, Feng YJ (2006) Quercetin reverses d-galactose induced neurotoxicity in mouse brain. Behav Brain Res 171:251–260
Maher P, Akaishi T, Abe K (2006) Flavonoid fisetin promotes ERK-dependent long-term potentiation and enhances memory. Proc Natl Acad Sci U S A 103:16568–16573
Malenka RC, Nicoll RA (1999) Long-term potentiation—a decade of progress? Science 285:1870–1874
McNaughton BL (1993) The mechanism of expression of long-term enhancement of hippocampal synapses: current issues and theoretical implications. Annu Rev Physiol 55:375–396
Middleton E Jr., Kandaswami C, Theoharides TC (2000) The effects of plant flavonoids on mammalian cells: implications for inflammation, heart disease, and cancer. Pharmacol Rev 52:673–751
Mizuno M, Yamada K, Maekawa N, Saito K, Seishima M, Nabeshima T (2002) CREB phosphorylation as a molecular marker of memory processing in the hippocampus for spatial learning. Behav Brain Res 133:135–141
Nguyen TT, Tran E, Nguyen TH, Do PT, Huynh TH, Huynh H (2004) The role of activated MEK-ERK pathway in quercetin-induced growth inhibition and apoptosis in A549 lung cancer cells. Carcinogenesis 25:647–659
Pande M, Flora SJ (2002) Lead induced oxidative damage and its response to combined administration of alpha-lipoic acid and succimers in rats. Toxicology 177:187–196
Patra RC, Swarup D, Dwivedi SK (2001) Antioxidant effects of alpha tocopherol, ascorbic acid and L-methionine on lead induced oxidative stress to the liver, kidney and brain in rats. Toxicology 162:81–88
PDRNS (2001) Quercetin. In: Physicians’ Desk Reference For Nutritional Supplements,1st Ed. Physicians’ Desk Reference (PDR), Demoines, Iowa/Medical Economics Data Production Company, Montvale, New Jersey, pp 390–392. Available from: <http://www.pdrhealth.com/drug_info/nmdrugprofiles/nutsupdrugs/que_0219.shtml>
Pu F, Mishima K, Irie K, Motohashi K, Tanaka Y, Orito K, Egawa T, Kitamura Y, Egashira N, Iwasaki K, Fujiwara M (2007) Neuroprotective effects of quercetin and rutin on spatial memory impairment in an 8-arm radial maze task and neuronal death induced by repeated cerebral ischemia in rats. J Pharmacol Sci 104:329–334
Reiter RJ (1995) Oxidative processes and antioxidative defense mechanisms in the aging brain. Faseb J 9:526–533
Rice-Evans CA, Miller NJ, Bolwell PG, Bramley PM, Pridham JB (1995) The relative antioxidant activities of plant-derived polyphenolic flavonoids. Free Radic Res 22:375–383
Ruan DY, Chen JT, Zhao C, Xu YZ, Wang M, Zhao WF (1998) Impairment of long-term potentiation and paired-pulse facilitation in rat hippocampal dentate gyrus following developmental lead exposure in vivo. Brain Res 806:196–201
Simon JA, Hudes ES (1999) Relationship of ascorbic acid to blood lead levels. Jama 281:2289–2293
Singh D, Chander V, Chopra K (2004) The effect of quercetin, a bioflavonoid on ischemia/reperfusion induced renal injury in rats. Arch Med Res 35:484–494
Spencer JP, Rice-Evans C, Williams RJ (2003) Modulation of pro-survival Akt/protein kinase B and ERK1/2 signaling cascades by quercetin and its in vivo metabolites underlie their action on neuronal viability. J Biol Chem 278:34783–34793
Sullivan M, Follis RH Jr., Hilgartner M (1951) Toxicology of podophyllin. Proc Soc Exp Biol Med 77:269–272
Tao X, Finkbeiner S, Arnold DB, Shaywitz AJ, Greenberg ME (1998) Ca2 + influx regulates BDNF transcription by a CREB family transcription factor-dependent mechanism. Neuron 20:709–726
Ternaux JP, Portalier P (2002) Effect of quercetine on survival and morphological properties of cultured embryonic rat spinal motoneurones. Neurosci Lett 332:33–36
Theoharides TC, Bielory L (2004) Mast cells and mast cell mediators as targets of dietary supplements. Ann Allergy Asthma Immunol 93:S24–34
Toscano CD, Hashemzadeh-Gargari H, McGlothan JL, Guilarte TR (2002) Developmental Pb2 + exposure alters NMDAR subtypes and reduces CREB phosphorylation in the rat brain. Brain Res Dev Brain Res 139:217–226
Toscano CD, McGlothan JL, Guilarte TR (2003) Lead exposure alters cyclic-AMP response element binding protein phosphorylation and binding activity in the developing rat brain. Brain Res Dev Brain Res 145:219–228
Villeda-Hernandez J, Barroso-Moguel R, Mendez-Armenta M, Nava-Ruiz C, Huerta-Romero R, Rios C (2001) Enhanced brain regional lipid peroxidation in developing rats exposed to low level lead acetate. Brain Res Bull 55:247–251
Viola H, Furman M, Izquierdo LA, Alonso M, Barros DM, de Souza MM, Izquierdo I, Medina JH (2000) Phosphorylated cAMP response element-binding protein as a molecular marker of memory processing in rat hippocampus: effect of novelty. J Neurosci 20:RC112
Wie MB, Ha HJ, Park SM (2000) Quercetin attenuates oxygen-glucose deprivation-induced neurotoxicity in cortical cultures. Soc Neurosci Abstr 26:2895
Youdim KA, Qaiser MZ, Begley DJ, Rice-Evans CA, Abbott NJ (2004) Flavonoid permeability across an in situ model of the blood-brain barrier. Free Radic Biol Med 36:592–604
Zelikoff JT, Parsons E, Schlesinger RB (1993) Inhalation of particulate lead oxide disrupts pulmonary macrophage-mediated functions important for host defense and tumor surveillance in the lung. Environ Res 62:207–222
Zucker RS (1989) Short-term synaptic plasticity. Annu Rev Neurosci 12:13–31
Acknowledgement
This work was supported by the National Basic Research Program of China (no. 2002CB512907), the National Nature Science Foundation of China (nos. 30630057; 30670554; 30670662; 30672290), Academia Sinica (No. KZCX3-SW-437), China Postdoctoral Science Foundation (No.20060400719), K. C. Wong Education Foundation of Hong Kong, and Anhui Provincial Natural Science Foundation (No.050430801).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hu, P., Wang, M., Chen, WH. et al. Quercetin relieves chronic lead exposure-induced impairment of synaptic plasticity in rat dentate gyrus in vivo. Naunyn-Schmied Arch Pharmacol 378, 43–51 (2008). https://doi.org/10.1007/s00210-008-0301-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00210-008-0301-z