Abstract
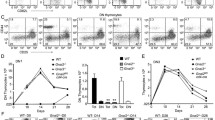
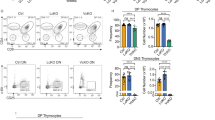
In the thymi of WT1-transgenic (Tg) mice with the 17AA+/KTS- spliced form of the Wilms tumor gene WT1 driven by the lck promoter, the frequencies of CD4-CD8- double-negative (DN) thymocytes were significantly increased relative to those in normal littermates. Of the 4 subsets of CD4-CD8- DN thymocytes, the DN1 (CD44+CD25-) subset increased in both frequency and absolute cell number, whereas the DN2 (CD44+CD25+) and DN3 (CD44-CD25+) subsets decreased, indicating the blocking of thymocyte differentiation from the DN1 to the DN2 subsets. Furthermore, CD4-CD8+ T-cell receptor (TCR) γδ T-cells increased in both frequency and absolute cell number in the spleen and peripheral blood of the WT1-Tg mice relative to those of normal littermates. The CD8 molecules of these CD4-CD8+ TCR78 T-cells were CD8aß, suggesting that they originated from the thymus. These results are the first direct evidence demonstrating that the WT1 gene is involved in the development and differentiation of T-lineage cells.IntJHematol. 2003;77:463-470.
Similar content being viewed by others
References
Call KM, Glaser T, Ito CY, et al. Isolation and characterization of a zinc finger polypeptide gene at the human chromosome 11 Wilms’ tumor locus.Cell. 1990;60:509–520.
Gessler M, Poustka A, Cavenee W, Neve RL, Orkin SH, Bruns GA. Homozygous deletion in Wilms tumours of a zinc-finger gene identified by chromosome jumping.Nature. 1990;343:774–778.
Menke AL, van der Eb AJ, Jochemsen AG. The Wilms’ tumor 1 gene: oncogene or tumor suppressor gene?Int Rev Cytol. 1998;181: 151–212.
Moore AW, McInnes L, Kreidberg J, Hastie ND, Schedl A. YAC complementation shows a requirement for WT1 in the development of epicardium, adrenal gland and throughout nephrogenesis.Development. 1999;126:1845–1857.
Miwa H, Beran M, Saunders GF. Expression of the Wilms’ tumor gene (WT1) in human leukemias.Leukemia. 1992;6:405–409.
Miyagi T, Ahuja H, Kubota T, Kubonishi I, Koeffler HP, Miyoshi I. Expression of the candidate Wilm’s tumor gene, WT1, in human leukemia cells.Leukemia. 1993;7:970–977.
Inoue K, Sugiyama H, Ogawa H, et al. WT1 as a new prognostic factor and a new marker for the detection of minimal residual disease in acute leukemia.Blood. 1994;84:3071–3079.
Brieger J, Weidmann E, Fenchel K, Mitrou PS, Hoelzer D, Bergmann L. The expression of the Wilms’ tumor gene in acute myelocytic leukemias as a possible marker for leukemic blast cells.Leukemia. 1994;8:2138–2143.
Menssen HD, Renkl HJ, Rodeck U, et al. Presence of Wilms’ tumor gene (wt1) transcripts and the WT1 nuclear protein in the majority of human acute leukemias.Leukemia. 1995;9:1060–1067.
Inoue K, Ogawa H, Sonoda Y, et al. Aberrant overexpression of the Wilms tumor gene (WT1) in human leukemia.Blood. 1997;89: 1405–1412.
Tamaki H, Ogawa H, Ohyashiki K, et al. The Wilms’ tumor gene WT1 is a good marker for diagnosis of disease progression of myelodysplastic syndromes.Leukemia. 1999;13:393–399.
Shichishima T, Okamoto M, Ikeda K, et al. HLA class II haplotype and quantitation of the WT1 gene in Japanese patients with paroxysmal nocturnal hemoglobinuria (PNH).Blood. 2002;100:22–28.
Park S, Schalling M, Bernard A, et al. The Wilms tumour gene WT1 is expressed in murine mesoderm-derived tissues and mutated in a human mesothelioma.Nat Genet. 1993;4:415–420.
Bruening W, Gros P, Sato T, et al. Analysis of the 11p13 Wilms’ tumor suppressor gene (WT1) in ovarian tumors.Cancer Invest. 1993;11:393–399.
Viel A, Giannini F, Capozzi E, et al. Molecular mechanisms possibly affecting WT1 function in human ovarian tumors.Int J Cancer. 1994;57:515–521.
Walker C, Rutten F, Yuan X, Pass H, Mew DM, Everitt J. Wilms’ tumor suppressor gene expression in rat and human mesothelioma.Cancer Res. 1994;54:3101–3106.
Rodeck U, Bossler A, Kari C, et al. Expression of the wt1 Wilms’ tumor gene by normal and malignant human melanocytes.Int J Cancer. 1994;59:78–82.
Ladanyi M, Gerald W Fusion of the EWS and WT1 genes in the desmoplastic small round cell tumor.Cancer Res. 1994;54: 2837–2840.
Amin KM, Litzky LA, Smythe WR, et al. Wilms’ tumor 1 susceptibility (WT1) gene products are selectively expressed in malignant mesothelioma.Am J Pathol. 1995;146:344–356.
Langerak AW, Williamson KA, Miyagawa K, Hagemeijer A, Versnel MA, Hastie ND. Expression of the Wilms’ tumor gene WT1 in human malignant mesothelioma cell lines and relationship to platelet-derived growth factor A and insulin-like growth factor 2 expression.Genes Chromosomes Cancer. 1995;12:87–96.
Silberstein GB, Van Horn K, Strickland P, Roberts CT Jr, Daniel CW. Altered expression of the WT1 Wilms tumor suppressor gene in human breast cancer.Proc Natl Acad Sci USA. 1997;94: 8132–8137.
Campbell CE, Kuriyan NP, Rackley RR, et al. Constitutive expression of the Wilms tumor suppressor gene (WT1) in renal cell carcinoma.IntJ Cancer. 1998;78:182–188.
Oji Y, Ogawa H, Tamaki H, et al. Expression of the Wilms’ tumor gene WT1 in solid tumors and its involvement in tumor cell growth.Jpn J Cancer Res. 1999;90:194–204.
Harada Y, Nonomura N, Nishimura K, et al. WT1 gene expression in human testicular germ-cell tumors.Mol Urol. 1999;3:357–363.
Menssen HD, Bertelmann E, Bartelt S, et al. Wilms’ tumor gene (WT1) expression in lung cancer, colon cancer and glioblastoma cell lines compared to freshly isolated tumor specimens.J Cancer Res Clin Oncol. 2000;126:226–232.
Miyoshi Y, Ando A, Egawa C, et al. High expression of Wilms’ tumor gene (WT1) predicts poor prognosis in breast cancer patients.J Clin Cancer Res. 2002;8:1167–1171.
Oji Y, Miyoshi S, Maeda H, et al. Overexpression of the Wilms’ tumor gene WT1 in de novo lung cancers.Int J Cancer. 2002;100: 304–308.
Yamagami T, Sugiyama H, Inoue K, et al. Growth inhibition of human leukemic cells by WT1 (Wilms tumor gene) antisense oligodeoxynucleotides: implications for the involvement of WT1 in leukemogenesis.Blood. 1996;87:2878–2884.
Algar EM, Khromykh T, Smith SI, Blackburn DM, Bryson GJ, Smith PJ. A WT1 antisense oligonucleotide inhibits proliferation and induces apoptosis in myeloid leukaemia cell lines.Oncogene. 1996;12:1005–1014.
Inoue K, Tamaki H, Ogawa H, et al. Wilms’ tumor gene (WT1) competes with differentiation-inducing signal in hematopoietic progenitor cells.Blood. 1998;91:2969–2976.
Tsuboi A, Oka Y, Ogawa H, et al. Constitutive expression of the Wilms’ tumor gene WT1 inhibits the differentiation of myeloid progenitor cells but promotes their proliferation in response to granulocyte colony-stimulating factor (G-CSF).Leuk Res. 1999;23: 499–505.
Phelan SA, Lindberg C, Call KM. Wilms’ tumor gene, WT1, mRNA is down-regulated during induction of erythroid and megakary-ocytic differentiation of K562 cells.Cell Growth Diff. 1994;5:677–686.
Sekiya M, Adachi M, Hinoda Y, Imai K, Yachi A. Downregulation of Wilms’ tumor gene (wt1) during myelomonocytic differentiation in HL60 cells.Blood. 1994;83:1876–1882.
Hosen N, Sonoda Y, Oji Y, et al. Very low frequencies of human normal CD34+ hematopoietic progenitor cells express the Wilms’ tumor gene WT1 at the levels similar to those in leukemia cells.Br J Haematol. 2002;116:409–420.
Nakagama H, Heinrich G, Pelletier J, Housman DE. Sequence and structural requirements for high-affinity DNA binding by the WT1 gene product.Mol Cell Biol. 1995;15:1489–1498.
Chaffin KE, Beals CR, Wilkie TM, Forbush KA, Simon MI, Perlmutter RM. Dissection of thymocyte signaling pathways byin vivo expression of pertussis toxin ADP-ribosyltranferase.EMBO J. 1990;9:3821–3829.
Shimizu C, Kawamoto H, Yamashita M, et al. Progression of T cell lineage restriction in the earliest subpopulation of murine adult thymus visualized by the expression oflck proximal promoter activity.Int Immunol. 2001;13:105–117.
Tsuboi A, Oka Y, Ogawa H, et al. Cytotoxic T-lymphocyte responses elicited to Wilms’ tumor gene WT1 product by DNA vaccination.J Clin Immunol. 2000;20:195–202.
Godfrey DI, Kennedy J, Suda T, Zlotnik A. A developmental pathway involving four phenotypically and functionally distinct subsets of CD3-CD4-CD8- triple-negative adult mouse thymocytes defined by CD44 and CD25 expression.J Immunol. 1993;150: 4244–4252.
Hoffman ES, Passoni L, Crompton T, et al. Productive T-cell receptor -chain gene rearrangement: coincident regulation of cell cycle and clonality during development in vivo.Genes Dev. 1996;10: 948–962.
Voll RE, Jimi E, Phillips RJ, et al. NF-κB activation by the pre-T cell receptor serves as a selective survival signal in T lymphocyte development.Immunity. 2000;13:677–689.
Marth JD, Peet R, Krebs EG, Perlmutter RM. A lymphocyte- specific protein-tyrosine kinase gene is rearranged and overex-pressed in the murine T cell lymphoma LSTRA.Cell. 1985;43: 393–404.
Wildin RS, Garvin AM, Pawar S, et al. Developmental regulation oflck gene expression in T lymphocytes.J Exp Med. 1991;173: 383–393.
Capone M, Hockett RD Jr, Zlotnik A. Kinetics of T cell receptor, -y, and 8 rearrangements during adult thymic development: T cell receptor rearrangements are present in CD44+CD25+ pro-T thymocytes.Proc Natl Acd Sci USA. 1998;95:12522–12527.
Livák F, Tourigny M, Schatz DG, Petrie HT. Characterization of TCR gene rearrangements during adult murine T cell development.J Immunol. 1999;162:2575–2580.
Shortman K, Wu L. Early T lymphocyte progenitors.Annu Rev Immunol. 1996;14:29–47.
Hewitt SM, Fraizer GC, Wu YJ, Rauscher FJ, Saunders GF. Differential functions of Wilms’ tumor geneWT1 splice isoforms in transcriptional regulation.J Biol Chem. 1996;271:8588–8592.
Davies R, Moore A, Schedl A, et al. Multiple roles for the Wilms’ tumor suppressor, WT1.Cancer Res. 1999;59(suppl):1747s-1751s.
Ellisen L, Carlesso N, Cheng T, Scadden DT, Haber DA. The Wilms tumor suppressor WT1 directs stage-specific quiescence and differentiation of human hematopoietic progenitor cells.EMBO J. 2001; 20:1897–1909.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Li, H., Oka, Y., Tsuboi, A. et al. The lck Promoter-Driven Expression of the Wilms Tumor Gene WT1 Blocks Intrathymic Differentiation of T-Lineage Cells. Int J Hematol 77, 463–470 (2003). https://doi.org/10.1007/BF02986614
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF02986614